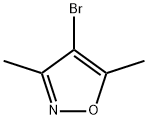
The general procedure for the synthesis of 4-bromo-3,5-dimethylisoxazole from 3,5-dimethylisoxazole is as follows:
1. Preparation of intermediate 105 (4-bromo-3,5-dimethyl-1,2-oxazole): DMF (53 mL) was added to a 250 mL round-bottomed flask fitted with a magnetic stirrer.
2. 3,5-dimethyl-1,2-oxazole (5.3 g, 54.57 mmol) and N-iodosuccinimide (11.659 g, 65.49 mmol) were sequentially added to the stirred DMF.
3. After completion of the addition, the reaction mixture was heated to 75°C and maintained for 3 hours.
4. Upon completion of the reaction, the mixture was diluted with ethyl acetate (100 mL).
5. The organic layer was washed sequentially with saturated NaHCO3 solution (100 mL), sodium thiosulfate solution (100 mL), water (200 mL) and brine (100 mL).
6. The organic layer was dried over anhydrous Na2SO4 and then concentrated under reduced pressure to remove the solvent to afford the target product 4-bromo-3,5-dimethyl-1,2-oxazole (6.1 g, 63.4% yield).