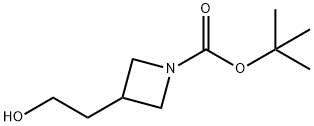
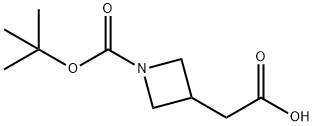
General procedure for the synthesis of tert-butyl 3-(2-hydroxyethyl)azetidine-1-carboxylate from N-Boc-3-azetidineacetic acid: 2-(1-(tert-butoxycarbonyl)azetidin-3-yl)acetic acid (3.91 g) was dissolved in tetrahydrofuran (THF, 18.17 mL), and the solution was cooled to 0 °C. The solution was then purified by stirring. Under stirring, ethylborane (1 M solution of THF, 54.5 mL) was added slowly and dropwise. The reaction mixture was stirred at 0 °C for 1 hour. Subsequently, water was added slowly and dropwise to quench the unreacted borane. The reaction mixture was diluted with ethyl acetate (EtOAc, 150 mL) and washed with deionized water (100 mL). The organic phase was dried with anhydrous sodium sulfate, filtered and concentrated under reduced pressure to give tert-butyl 3-(2-hydroxyethyl)azetidine-1-carboxylate as a clear oil in 100% yield. The product was confirmed by 1H NMR (CDCl3): δ 4.0 (t, 2H), 3.60 (m, 4H), 2.65 (m, 1H), 1.85 (m, 2H), 1.43 (s, 9H).