Synthesis
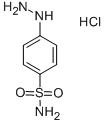
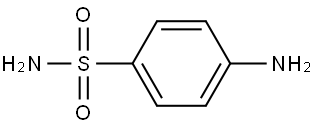
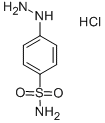
The general procedure for the synthesis of p-hydrazinylbenzenesulfonamide hydrochloride from crystalline sulfanilamide was as follows: firstly, 4'-methylacetophenone was reacted with succinic anhydride in the presence of diisopropanolamine (LDA) and tetrahydrofuran (THF) for 1 hr at -78 °C to afford p-tolyl-4,6-dioxohexanoic acid in 66% yield and the product was a white solid. Subsequently, the sulfonamide was reacted with sodium nitrite (NaNO2) and concentrated hydrochloric acid for 30 min at 0-4 °C, followed by the addition of tin(II) chloride (SnCl2), and the reaction was continued for 4 h at 0 °C to produce 4-sulfonylaminophenylhydrazine hydrochloride in 55% yield, the product being a pale yellow solid. Finally, p-tolyl-4,6-dioxohexanoic acid was reacted with 4-sulfonylaminophenylhydrazine hydrochloride in a methanolic solution of triethanolamine (TEA) at room temperature for 16 h to give compound 30c, 3-[1-{p-(p-(sulfonylamino)phenyl}-5-p-tolyl-1H-pyrazol-3-yl]propanoic acid in 76% yield and the product was a yellow solid.
References
[1] Journal of Medicinal Chemistry, 2007, vol. 50, # 18, p. 4444 - 4452
[2] Organic Letters, 2012, vol. 14, # 19, p. 5030 - 5033,4
[3] Journal of Heterocyclic Chemistry, 2018, vol. 55, # 4, p. 913 - 922
[4] Patent: US2007/292352, 2007, A1. Location in patent: Page/Page column 45-46
[5] Patent: US2003/236294, 2003, A1. Location in patent: Page 9