Description
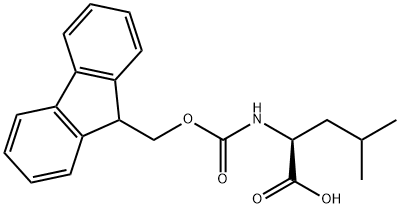
Fmoc-Leu-OH is isomeric with Fmoc-Ile-OH. Like isoleucine, leucine has branching in the sidechain. The branching is at the gamma carbon instead of the beta carbon as in isoleucine. It is used in chemical and peptide syntheses.
Chemical Properties
white to light yellow crystal powde
Application
Fmoc-Leu-OH can be used as a reactant to synthesize:
Various oligopeptides by reacting with functionalized α-amino acid hydrochloride salts.
A cyclic depsipeptide sansalvamide A, a natural product found in marine fungus.
Streptocidin A?D, decapeptide antibiotics naturally found in Streptomyces?sp. Tü 6071.
Coumaroyl dipeptide amide that can be used for cosmetic applications.
Uses
Fmoc-L-Leu-OH, is an amino acid derivative, used in peptide chemistry. It is also one of the novel PPARγ ligands that can activate PPARγ in different ways, that reduces osteoclasts differentiation, and thus are better therapeutic targets in diabetes than traditional antidiabetic drugs.
Preparation
Fmoc-L-leucine is prepared by reacting L-leucine with 9-fluorenylmethoxycarbonyl chloride.
reaction suitability
reaction type: Fmoc solid-phase peptide synthesis
Biological Activity
Anti-inflammatory agent; increases intracellular Ca 2+ levels.
Biochem/physiol Actions
PPARγ ligand that induces insulin sensitization, but not adipogenesis.
Synthesis
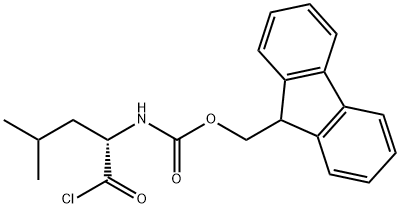
The general procedure for the synthesis of Fmoc-L-leucine and compound (CAS:139551-97-6) using compound (CAS:103321-59-1) as starting material was as follows:Example 22 Synthesis of N-[(9H-fluoren-9-ylmethoxy)carbonyl]-L-leucine amide (NPC 15528). To a 10 mL THF solution containing 3.5 mL (10 mmol) of N-[(9H-fluoren-9-ylmethoxy)carbonyl]-L-leucine chloride, 1.26 mL (20 mmol) of a 29.6% ammonia solution was slowly added at room temperature. The reaction mixture was stirred for 30 min, then diluted with 100 mL of water and extracted with ethyl acetate (5 x 20 mL). The organic layers were combined and washed sequentially with 10% potassium carbonate solution, water and brine, then dried and concentrated. The product was recrystallized by ethanol to give 1.26 g (36% yield) of N-(9-fluorenylmethoxycarbonyl)-L-leucine amide as a colorless solid with a melting point of 79-80 °C.
References
[1] Patent: US5079260, 1992, A