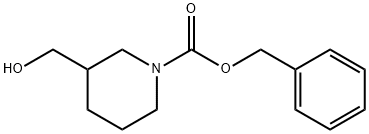
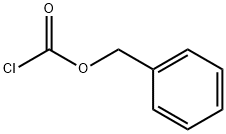
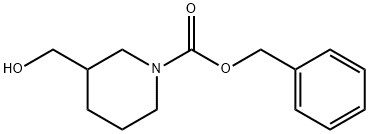
Triethylamine (0.51 g, 5 mmol) was added to a frozen solution of 3-hydroxymethylpiperidine (0.61 g, 5 mmol) in dichloromethane (10 mL), followed by slow dropwise addition of benzyl chloroformate (0.88 g, 5 mmol). The reaction mixture was gradually warmed to room temperature and stirred continuously overnight. After completion of the reaction, dichloromethane (50 mL) was added to dilute the reaction mixture, and the organic layer was washed sequentially with 5% hydrochloric acid solution (2 × 20 mL), saturated sodium bicarbonate solution (20 mL), and saturated sodium chloride solution (20 mL). The organic layer was dried over anhydrous sodium sulfate, filtered to remove the desiccant, and concentrated under reduced pressure to remove the solvent. The crude product was purified by fast column chromatography (silica gel, ethyl acetate/hexane as eluent) to afford the target compound 3-methanol-N-benzyloxycarbonylpiperidine (1.21 g, 96% yield) as a colorless oil.1H NMR (CDCl3) δ: 1.20-1.83 (m, 5H), 2.78-3.20 (m, 2H), 3.50 (s, 2H), 3.69-4.05 (m, 2H), 5.14 (s, 2H), 7.25-7.40 (m, 5H).