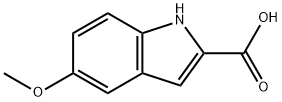
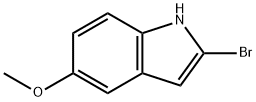
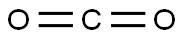To a solution of 2-bromo-5-methoxy-1H-indole (1.0 g, 4.4 mmol, 1.0 eq.) in anhydrous THF (20 mL) was slowly added 2M isopropylmagnesium chloride (i-PrMgCl) in THF solution (2.2 mL, 4.4 mmol, 1.0 eq.) at 0 °C. The reaction mixture was stirred at this temperature for 5 minutes, followed by the dropwise addition of 2.5 M hexane solution (3.5 mL, 8.8 mmol, 2.0 eq.) of n-butyllithium (n-BuLi) over a period of 5 minutes, controlling the reaction temperature to not exceed 20 °C. The resulting mixture was continued to be stirred at 0 °C for 30 min and then passed through dry carbon dioxide gas (0.20 g, 4.4 mmol, 1.0 eq.). The reaction system was slowly warmed to 20 °C over 0.5 h and the reaction was quenched with deionized water (6 mL). After stirring for 10 min, the organic and aqueous phases were separated and the aqueous phase was extracted once with ethyl acetate (10 mL). The organic phases were combined, the suspension was returned to room temperature, filtered through a silica gel pad (0.5 cm) and eluted with ethyl acetate (10 mL). The filtrate was concentrated and the residue was purified by silica gel column chromatography (eluent: petroleum ether/ethyl acetate = 3:1) to afford the brown solid product 5-methoxyindole-2-carboxylic acid (0.68 g, 80% yield) with a melting point of 199-201 °C. The structure of the product was confirmed by 1H-NMR (600 MHz, DMSO-d6) and 13C-NMR (151 MHz, DMSO-d6).