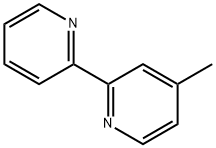
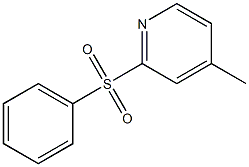
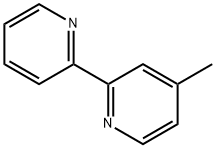
Example 2 Synthesis of 4-methyl-2-(2'-pyridinyl)pyridine: Tetrahydrofuran (10 ml) and isopropylmagnesium chloride (2.0 M, 7.7 ml, 15.5 mmol) were added to a 50 ml flask under nitrogen protection. Subsequently, a solution prepared from 2-bromopyridine (2.45 g, 15.5 mmol) dissolved in tetrahydrofuran (3 ml) was added slowly and dropwise over 10 min. The reaction mixture was stirred for 1 hour and then a solution prepared from 4-methyl-2-benzenesulfonylpyridine (3.00 g, 12.9 mmol) dissolved in tetrahydrofuran (5 ml) was added dropwise over 10 minutes. The reaction mixture was continued to be stirred at room temperature for 5 h. Isopropanol (1 ml) was added at the termination of the reaction. The reaction mixture was poured into water and extracted with ethyl acetate (15 ml x 2). The organic phases were combined, concentrated and purified by silica gel column chromatography to afford the target product 4-methyl-2,2'-bipyridine (1.51 g, 89% yield, based on 4-methyl-2-phenylsulfonylpyridine) as a white solid. The product was characterized by 1H-NMR (CDCl3): δ 2.36 (s, 3H), 7.06 (d, 1H, J = 5.0 Hz), 7.20-7.25 (m, 1H), 7.69-7.74 (m, 1H), 8.16 (s, 1H), 8.32 (d, 1H, J = 8.0 Hz), 8.46 (d, 1H, J = 4.6 Hz) , 8.57-8.63 (m, 1H).