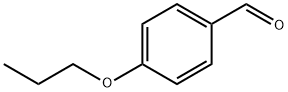
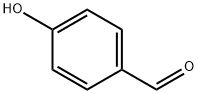
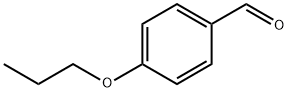
Step C: Synthesis of 3-chloro-4-propoxybenzaldehyde: A mixture of 4-hydroxybenzaldehyde (0.5 g, 4.1 mmol), 1-bromopropane (0.3 ml), and K2CO3 (0.69 g, 5 mmol) in anhydrous DMF (5 ml) was stirred and refluxed for 1 hour. After completion of the reaction, it was diluted to 100 ml with EtOAc and washed with H2O. The organic layer was separated, dried with MgSO4 and filtered. The filtrate was evaporated to dryness to give 4-propoxybenzaldehyde (0.55 g, 82.3%) as a yellow oil.1H-NMR (CDCl3) δ: 1.0 (t, 3H, J=7.41Hz); 1.7-2.0 (m, 2H); 3.95 (t, 2H, J=6.57Hz); 6.95 (d, 2H, J=8.73Hz); 8.00 (d, 2H, J=8.76Hz). To a stirred solution of 4-propoxybenzaldehyde (0.5 g, 3.07 mmol) in anhydrous DMF (3 ml) was added N-chlorosuccinimide (NCS, 0.5 g, 3.73 mmol) and the mixture was stirred at room temperature overnight. After completion of the reaction, it was diluted to 100 ml with EtOAc and washed with NaHCO3 solution (100 ml). The organic layer was separated, dried with MgSO4 and filtered. The filtrate was evaporated to dryness to give the crude product 3-chloro-4-propoxybenzaldehyde (0.59 g; 97%) as a light yellow oil.1H-NMR (CDCl3) δ: 9.81 (s, 1H); 7.87 (d, 1H, J=2.01 Hz); 7.71 (dd, 1H, J=2.02-8.47Hz); 6.98 (d, 1H, J= 8.47 Hz); 1.82-1.91 (m, 2H); 4.1 (t, 2H, J=6.3 Hz); 1.1 (t, 3H, J=7.4 Hz).