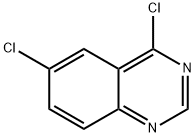
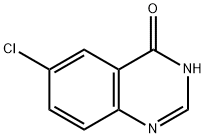
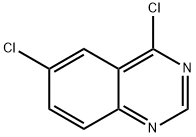
General procedure for the synthesis of 4,6-dichloroquinazoline from 6-chloroquinazolin-4-one: 6-chloroquinazolin-4-one (400 mg, 2.21 mmol, 1.0 eq.), phosphoryl chloride (1.99 mL, 21.4 mmol, 9.64 eq.) and triethylamine (0.99 mL, 7.11 mmol, 3.21 eq.) were mixed under nitrogen protection in a reaction flask. The reaction mixture was stirred and refluxed for 2.5 hours at room temperature. Upon completion of the reaction, the solvent was removed by rotary evaporation and the residue was recrystallized twice with toluene to give a brown solid. The solid was dissolved in dichloromethane (25 mL) and the organic phase was washed twice with saturated ammonium chloride solution (25 mL). The organic layer was dried over anhydrous sodium sulfate and concentrated to give a brown solid. The crude product was purified by silica gel column chromatography with an eluent of 9:1 to 3:1 hexane/ethyl acetate to give 4,6-dichloroquinazoline (300 mg) as an off-white solid in 68% yield. The structure of the product was confirmed by 1H NMR (400 MHz, DMSO-d6): δ 9.16 (s, 1H), 8.33 (s, 1H), 8.17 (apparent t, 2H, J = 7 Hz).