Synthesis
The general procedure for the synthesis of pyrimidine-4(3H)-thione from pyrimidin-4(1H)-one is as follows:
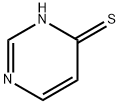
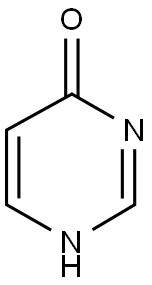
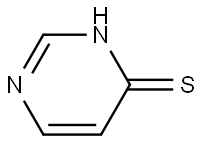
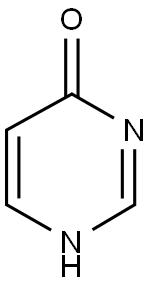
Step a: Preparation of 4-mercaptopyrimidine
4-Hydroxypyrimidine (1 g) and diphosphorus pentasulfide (2.32 g) were dissolved in 15 ml of pyridine, stirred and heated to reflux. After refluxing the reaction mixture for 3 hours, it was cooled to room temperature and 30 ml of water was added. Subsequently, the reaction mixture was concentrated under reduced pressure. The concentrated mixture was extracted with ethyl acetate (3 x 30 ml), the organic phases were combined, washed with water, dried over anhydrous MgSO4, filtered and the solvent was evaporated under reduced pressure to give the yellow solid product 4-mercapto-pyrimidine (0.42 g, 36% yield). The product did not need further purification and could be used directly in the subsequent reaction.