Description
AZD1152-HQPA (722544-51-6) is a potent and highly selective inhibitor of Aurora B.1Aurora B Ki= 0.36 nM; Aurora A Ki= 1,369 nM.2It inhibits the growth of tumors in multiple cancer models.3-6Excessive ROS generation7and upregulated tumor suppressor miRNAs8have been proposed as a novel mechanisms of cytotoxicity of AZD1152-HQPA.
Uses
AZD1152-HQPA is a a highly selective inhibitor of aurora-B kinase. AZD1152-HQPA has been shown to inhibit osteosarcoma cell line U2-OS cells proliferation. AZD1152-HQPA was also shown to be an effective apoptosis-inducing agent in acute myeloid leukemia cell lines and primary acute myeloid leukemia cultures.
Uses
AZD1152-HQPA has been used:
- for the dose dependent inhibition of aurora kinase B (AURKB)
- as a component to identify that, replication timing regulatory factor 1 (RIF1) and protein phosphatase 1 (PP1) are critical for the regulation of abscission timing in human cells
- as a component to study the relevance of AURKB as a cancer therapeutic target
Definition
ChEBI: AZD-1152 is a member of the of quinazolines that is 4-aminoquinazolin-7-ol in which the amino group at position 4 has been substituted by a 5-[2-(3-fluoroanilino)-2-oxoethyl]-1H-pyrazol-3-yl group, while the hydroxy group at position 7 has been converted into the corresponding 3-[ethyl(2-hydroxyethyl)aminopropyl ether. It has a role as an antineoplastic agent and an Aurora kinase inhibitor. It is a member of quinazolines, a secondary carboxamide, a tertiary amino compound, a secondary amino compound, a member of pyrazoles, a primary alcohol, a member of monofluorobenzenes and an anilide.
Biochem/physiol Actions
AZD1152-HQPA is the active metabolite of AZD-1152, a potent selective Aurora Kinase B inhibitor. AZD1152 is 50-fold selective for Aurora kinase B over Aurora kinase C and over 1000-fold selective for Aurora kinase B over Aurora Kinase A. (IC50s: aurora-A, 1,369 nmol/L; aurora-B, 0.36 nmol/L; aurora-C, 17.0 nmol/L). It is converted in plasma to the active form AZD1152-HQPA, which has been shown to have antineoplastic activity in a variety of animal models and human cancer cell lines.
Synthesis
The general procedure for the synthesis of 2-(5-((7-(3-(3-(ethyl(2-hydroxyethyl)amino)propoxy)quinazolin-4-yl)amino)-1H-pyrazol-3-yl)-N-(3-fluorophenyl)acetamide, using the compound (CAS:722544-46-9) and N-ethyl ethanolamine, was carried out as follows: firstly, in an inert atmosphere (e.g., protected by nitrogen), 2-(3-(3-(3-(3-chloropropoxy)quinazolin-4-yl)amino}-1H-pyrazole-5-yl)-N-(3-fluorophenyl)acetamide was synthesized as follows. {[7-(3-chloropropoxy)quinazolin-4-yl]amino}-1H-pyrazol-5-yl)-N-(3-fluorophenyl)acetamide and N-ethyl ethanolamine were dissolved in N,N-dimethylacetamide under an inert atmosphere (e.g., nitrogen protection). Subsequently, the reaction mixture was heated to 90°C under stirring and maintained at this temperature for 12-16 hours (optimally 12 hours). Upon completion of the reaction, the mixture was cooled to about 85°C and water was slowly added to maintain the temperature between 80-85°C. The temperature of the mixture was adjusted to 80°C and a crystal seed with an expected yield of about 1% was added to induce crystallization. Over the next about 20 hours, the mixture was slowly cooled to 20°C to ensure that the product precipitated in the desired crystalline form and that the crystals were of suitable size for filtration. The product was collected by filtration, washed sequentially with a water/N,N-dimethylacetamide mixture and acetonitrile, and subjected to appropriate de-liquefaction to give the product in hydrated form. Subsequently, the filter cake was slurried with warm acetonitrile (about 40°C) for about 2 hours, filtered again, washed with acetonitrile, and finally dried under vacuum or nitrogen flow to give virtually anhydrous 2-{3-[(7-{3-[ethyl(2-hydroxyethyl)amino]propoxy}- quinazolin-4-yl)amino]-1H-pyrazol-5-yl}-N-(3-fluorophenyl)acetamide as an off-white solid in yields of 85-90%. The product was confirmed by 1H-NMR (DMSO-d6) and mass spectrometry (+ve ESI), the characteristic signals and molecular ion peaks were as follows: 1H-NMR (DMSO-d6): δ 10.55 (s, 1H), 9.45 (br s, 1H), 8.98 (s, 1H), 8.8 (d, 1H), 7.63 (m, 1H), 7.47 (pr d 1H), 7.37 (m, 2H), 7.32 (d, 1H), 6.9 (m, 1H), 6.77 (s, 1H), 4.32 (t, 2H), 3.83 (br s, 2H), 3.76 (t, 2H), 3.35 (m, 2H), 3.25 (m, 4H), 2.25 (m, 2H), 1.25 (t, 3H); MS (+ve ve ESI): m/z 508.4 (M + H)+.
References
[1] ANDREW A. MORTLOCK. Discovery, Synthesis, and in Vivo Activity of a New Class of Pyrazoloquinazolines as Selective Inhibitors of Aurora B Kinase[J]. Journal of Medicinal Chemistry, 2007, 50 9: 2213-2224. DOI:
10.1021/jm061335f[2] ROBERT W WILKINSON. AZD1152, a selective inhibitor of Aurora B kinase, inhibits human tumor xenograft growth by inducing apoptosis.[J]. Clinical Cancer Research, 2007, 13 12: 3682-3688. DOI:
10.1158/1078-0432.ccr-06-2979[3] ADEDAYO OKE. AZD1152 rapidly and negatively affects the growth and survival of human acute myeloid leukemia cells in vitro and in vivo.[J]. Cancer research, 2009: 4150-4158. DOI:
10.1158/0008-5472.can-08-3203[4] ARIHIRO AIHARA. The selective Aurora B kinase inhibitor AZD1152 as a novel treatment for hepatocellular carcinoma[J]. Journal of Hepatology, 2010, 52 1: Pages 63-71. DOI:
10.1016/j.jhep.2009.10.013[5] NAOKI MORI . Effects of AZD1152, a selective Aurora B kinase inhibitor, on Burkitt’s and Hodgkin’s lymphomas[J]. Biochemical pharmacology, 2011, 81 9: Pages 1106-1115. DOI:
10.1016/j.bcp.2011.02.010[6] BARBARA A HELFRICH. Barasertib (AZD1152), a Small Molecule Aurora B Inhibitor, Inhibits the Growth of SCLC Cell Lines In Vitro and In Vivo.[J]. Molecular Cancer Therapeutics, 2016, 15 10: 2314-2322. DOI:
10.1158/1535-7163.mct-16-0298[7] ALI ZEKRI. Reactive oxygen species generation and increase in mitochondrial copy number: new insight into the potential mechanism of cytotoxicity induced by aurora kinase inhibitor, AZD1152-HQPA.[J]. Anti-Cancer Drugs, 2017, 28 8: 841-851. DOI:
10.1097/cad.0000000000000523[8] ALI ZEKRI. The Potential Contribution of microRNAs in Anti-cancer Effects of Aurora Kinase Inhibitor (AZD1152-HQPA)[J]. Journal of Molecular Neuroscience, 2018, 65 4: 444-455. DOI:
10.1007/s12031-018-1118-y
![1H-Pyrazole-3-acetamide, 5-[[7-[3-[ethyl(2-hydroxyethyl)amino]propoxy]-4-quinazolinyl]amino]-N-(3-fluorophenyl)- Structure](https://www.chemicalbook.com/CAS/GIF/722544-51-6.gif)


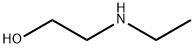
![1H-Pyrazole-3-acetamide, 5-[[7-[3-[ethyl(2-hydroxyethyl)amino]propoxy]-4-quinazolinyl]amino]-N-(3-fluorophenyl)-](/CAS/GIF/722544-51-6.gif)

