Synthesis
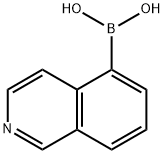
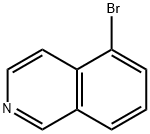
To a solution of 5-bromoisoquinoline (1.46 g, 7.0 mmol) in THF (20 mL) prepared according to the literature method (Brown, W. D et al., Synthesis 2002, 83), triisopropyl borate (2.4 mL, 10.5 mmol) and tertiary-butyllithium (1.7 M, 9.5 mL) were sequentially added at -78 °C. The reaction mixture was slowly warmed to room temperature and stirred overnight. Upon completion of the reaction, the reaction was quenched with 1N HCl (10 mL) and a solid product was obtained after decantation of the THF layer. The solid was identified as 5-isoquinolineboronic acid (1.0 g, 82% yield). The mass spectrum (ESI) showed the (M + 1) peak as 174.12.
References
[1] Patent: WO2005/72732, 2005, A1. Location in patent: Page/Page column 61
[2] Patent: WO2008/6480, 2008, A1. Location in patent: Page/Page column 5-6