Synthesis
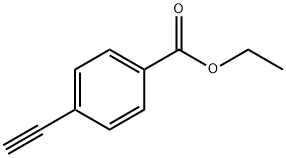
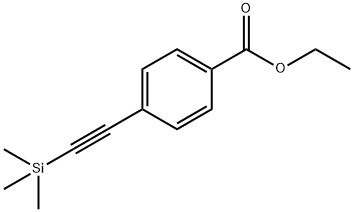
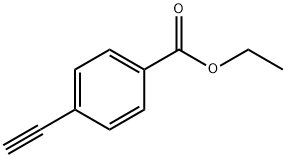
The general procedure for the synthesis of ethyl 4-alkynylbenzoate from the compound (CAS:150969-54-3) is as follows: firstly, the desired aryl-substituted terminal acetylene (5) was prepared using the Sonogashira reaction [1] as a precursor for the synthesis of betulinic acid 1,2,3-triazole library. Some of the acetylene (5a, 5b, 5c, 5g) can be purchased directly from commercial sources. This was done as follows: under argon protection, iodide 6 (500 mg) was reacted with trimethylmethylsilylacetylene (1.5 eq.) in the presence of Pd(PPh3)2Cl2 (2 mol%), CuI (4 mol%), and triethylamine (0.5 mL) in dry DMF at room temperature to produce intermediate 7. Subsequently, intermediate 7 was formed by reaction of iodide 6 with trimethylsilylacetylene (1.5 eq.) dissolved in anhydrous methanol (4.0 mL ) in anhydrous methanol (4.0 mL), the demethylsilylation reaction was achieved by treating intermediate 7 with a solution of K2CO3 (1 equiv.) to ultimately yield terminal acetylene 5.
References
[1] Chemistry - A European Journal, 2011, vol. 17, # 34, p. 9320 - 9325
[2] European Journal of Medicinal Chemistry, 2015, vol. 102, p. 93 - 105
[3] Patent: WO2009/149381, 2009, A2. Location in patent: Page/Page column 32
[4] Journal of Polymer Science, Part A: Polymer Chemistry, 2011, vol. 49, # 1, p. 211 - 224
[5] Journal of the American Chemical Society, 2015, vol. 137, # 1, p. 413 - 419