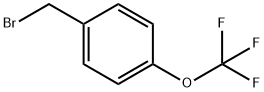
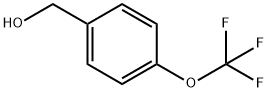
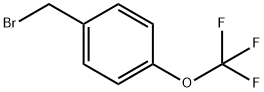
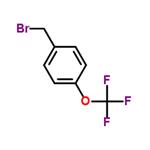
Uses
4-Trifluoromethoxybenzyl Bromide is a useful synthetic intermediate. It is used to prepare (nitro)[(trifluoromethoxy)benzyloxy]dihydroimidazo[2,1-b][1,3]oxazines with antitubercular activities. It is also used to synthesize tetrahydronaphthalenols with anti-allergic activities.
Chemical Properties
Colorless to light yellow liqui
Uses
4-(Trifluoromethoxy)benzyl bromide may be used in the synthesis of bioreductive drug, (6
S)-2-nitro-6-{[4-(trifluoromethoxy)benzyl]oxy}-6,7-dihydro-5
H-imidazo[2,1-
b][1,3]oxazine (PA-824).
General Description
Polymerizations of 4-(trifluoromethoxy)benzyl bromide, via Friedel-Crafts polymerization using aluminum chloride as a catalyst has been reported.
Synthesis
Synthesis of 1-(bromomethyl)-4-(trifluoromethoxy)benzene: Phosphorus tribromide (6 g, 22.2 mmol) was slowly added dropwise to a solution of (4-(trifluoromethoxy)phenyl)methanol (3.6 g, 18.8 mmol) in dichloromethane (60 mL) at 0-10 °C and stirred for 30 min. The reaction mixture was then continued to stir at room temperature for 3 hours. Upon completion of the reaction, the reaction was terminated by the addition of water (30 mL) and the mixture was washed sequentially with aqueous sodium bicarbonate (2 x 30 mL) and brine (30 mL). The organic layer was dried with anhydrous sodium sulfate and concentrated under reduced pressure to give 4-trifluoromethoxybenzyl bromide (3.5 g, 66% yield) as a yellow oil.
References
[1] Patent: US2007/270434, 2007, A1. Location in patent: Page/Page column 17
[2] J. Gen. Chem. USSR (Engl. Transl.), 1965, vol. 35, p. 1631 - 1637
[3] Zhurnal Obshchei Khimii, 1965, vol. 35, p. 1628 - 1636
[4] Journal of Medicinal Chemistry, 2005, vol. 48, # 2, p. 556 - 568
[5] Organometallics, 2014, vol. 33, # 21, p. 5940 - 5943