Synthesis
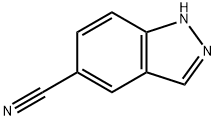
General procedure for the synthesis of indazole-5-carbonitrile from 4-fluoro-3-formylbenzonitrile: 4-fluoro-3-formylbenzonitrile (25 g, 16.78 mmol) was dissolved in 100 mL of 85% hydrazine hydrate. The reaction mixture was stirred at room temperature for 24 hours. After completion of the reaction, the product was purified by column chromatography to afford 1H-indazole-5-carbonitrile (2.1 g, 87% yield). The structure of the product was confirmed by 1H NMR (400 MHz, DMSO-d6) and mass spectrometry (ESI): 1H NMR δ 13.60 (s, 1H), 8.42 (s, 1H), 8.27 (s, 1H), 7.73 (d, J = 8.6 Hz, 1H), 7.67 (d, J = 8.6 Hz, 1H); MS m/z (ESI): 144 [M + H]+.
References
[1] Patent: EP3205650, 2017, A1. Location in patent: Paragraph 0106; 0345
[2] Bioorganic and Medicinal Chemistry Letters, 2009, vol. 19, # 15, p. 4191 - 4195
[3] Synthetic Communications, 1997, vol. 27, # 7, p. 1199 - 1207
[4] Patent: US6376499, 2002, B1
[5] Patent: US6610692, 2003, B1