Synthesis
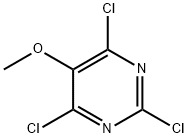
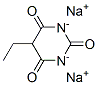
Step 2: 2,4,6-Trichloro-5-methoxypyrimidine-6-hydroxy-5-methoxy-1H-pyrimidine-2,4-dione sodium salt (21 mmol) was suspended in phosphoryl chloride (20 mL). The mixture was dispensed in two 20 mL microwave reaction vials. The reaction mixture was heated at 130-140 °C (pressure about 10-12 bar) for 30 min (significant pressure rise was observed) using microwave radiation. After the reaction mixtures were cooled, they were carefully combined and slowly poured into water preheated to about 40°C. The resulting mixture was extracted twice with ethyl acetate, the organic phases were combined, dried over anhydrous sodium sulfate, filtered and concentrated under reduced pressure to give 2,4,6-trichloro-5-methoxypyrimidine as a yellow/brown crystalline solid (3.75 g, 84% yield).1H NMR (CDCl3): δ 3.98 (s, 3H).