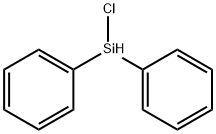
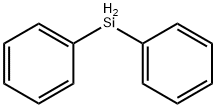
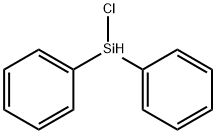
Physical properties
bp 140–145°C/7 mmHg (99–101°C/1 mmHg;
83–85°C/0.4 mmHg); d 1.137 g cm3; n 1.5845.
Uses
Chlorodiphenylsilane is used in Medicine field, electronics industry, polymer materials. It is used in reduction of alcohols.
Uses
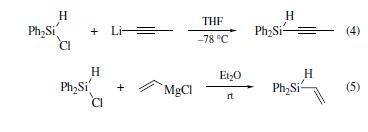
Organolithium and Grignard reagents react with chlorodiphenylsilane to produce substituted alkyl- or aryldiphenylsilanes (eqs 4 and 5).
Preparation
chlorodiphenylsilane may be prepared in
high yield by treatment of diphenylsilane with triphenylmethyl
chloride in refluxing benzene or with PCl5 at rt in CCl4.
Synthesis
General procedure for the synthesis of diphenylchlorosilane from diphenylsilane: A mixture of 18.44 g (0.1 mol) of diphenylsilane (Ph2SiH2), 26.9 g (0.2 mol) of anhydrous copper chloride (CuCl2), 0.92 g (0.0048 mol) of anhydrous cuprous iodide (CuI), 80 g of ceramic balls, and 40 mL of diethyl ether was placed in a reactor at 50 °C using a mechanical stirrer at 400 rpm. The reaction was continued at room temperature for 2 hours. After completion of the reaction, the mixture was filtered and the filtrate was collected and concentrated. The concentrated residue was distilled under reduced pressure to give diphenylchlorosilane (Ph2SiHCl) (20.24 g, 92.6% yield) as a colorless liquid. Boiling point: 117 °C (210 Pa).1H NMR (δ, ppm): 5.82 (s, 1H, SiH), 7.47-7.76 (m, 10H, Si(C6H5)2).13C NMR (δ, ppm): 128.36, 131.10, 131.85, 134.52 (m, 12C, Si(C6H5)2). 29Si NMR (δ, ppm): -5.94 (s, 1Si, Ph2SiHCl).
References
[1] Journal of the Chemical Society - Perkin Transactions 1, 1998, # 3, p. 467 - 475
[2] Journal of Organometallic Chemistry, 2014, vol. 769, p. 29 - 33
[3] Patent: CN103880874, 2017, B. Location in patent: Paragraph 0043; 0044
[4] Journal of the Chemical Society - Perkin Transactions 1, 1998, # 17, p. 2687 - 2700
[5] J. Gen. Chem. USSR (Engl. Transl.), 1971, vol. 41, p. 1083 - 1087