Synthesis
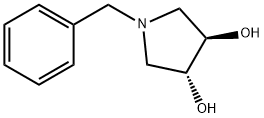
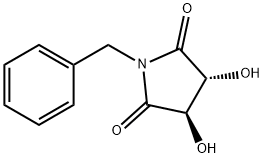
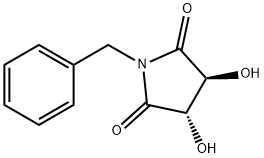
General procedure for the synthesis of (3R,4R)-1-benzyl-3,4-dihydroxypyrrolidine-2,5-dione from (3R,4R)-1-benzyl-3,4-pyrrolidinediol: a tetrahydrofuran solution of borane (1.0 M, 17.19 mL, 180.99 mmol) was added slowly and dropwise to a tetrahydrofuran solution (1.0 M, 17.19 mL, 180.99 mmol) of (3R,4R)-1-benzyl-3,4-dihydroxy pyrrolidine-2,5-dione (10.0 g, 45.24 mmol) in a solution of tetrahydrofuran (200 mL). After dropwise addition, the reaction mixture was slowly heated to 70°C and kept at this temperature for 3 hours. Upon completion of the reaction, the mixture was cooled to 0°C and the reaction was quenched by the dropwise addition of methanol (50 mL), noting that a strong gas release occurs at this point. Subsequently, the mixture was slowly warmed to room temperature and stirring was continued for 1 hour. The reaction mixture was concentrated under vacuum and the crude product was purified by silica gel column chromatography using a linear gradient elution of ethyl acetate in petroleum ether to afford 7.73 g (88.5% yield) of the yellow crystalline product (3R,4R)-1-benzyl-3,4-pyrrolidinediol. Specific optical rotation (MeOH, 0.5%): +22.800°; 1H NMR (400 MHz, DMSO-d6) δ 7.51-7.49 (2H, m), 7.39-7.37 (3H, m), 5.45 (1H, d, J = 4.4 Hz, OH), 5.20 (1H, d, J = 5.2 Hz, OH), 4.15- 4.11 (1H, m), 4.04-3.94 (2H, m), 3.90-3.85 (1H, m), 3.48-3.43 (1H, m), 3.10-3.06 (1H, m), 2.97-2.93 (1H, m), 2.89-2.86 (1H, m); 13C NMR (100 MHz, DMSO-d6) δ 132.86, 128.97, 128.51, 127.73, 76.27, 65.47, 63.24; LC-MS (ESI) m/z Calculated value C11H15NO2 [M + H]+: 193.11, measured value: 194.20.
References
[1] Bioorganic and Medicinal Chemistry Letters, 2017, vol. 27, # 12, p. 2818 - 2823
[2] Synlett, 2009, # 12, p. 1945 - 1948