Synthesis
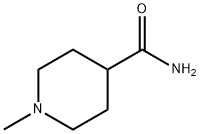
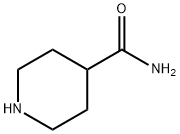
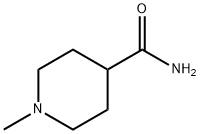
Piperidine-4-carboxamide (10 g, 78 mmol) was dissolved in distilled water (100 mL) and 37% aqueous formaldehyde solution (7.6 mL, equivalent to 2.81 g HCHO, 93.6 mmol) was added. Under argon protection, wet 10% Pd/C catalyst (8 spatula scoops) was added and the mixture was hydrogenated for 43 h at 25 °C and 50 psi hydrogen pressure. Upon completion of the reaction, the catalyst was removed by diatomaceous earth filtration and the catalyst was washed with water and methanol. The filtrates were combined and evaporated to dryness. The residue was purified by silica gel column chromatography (60 x 5 cm) to afford 1-methylpiperidine-4-carboxamide (7.15 g, 64% yield) using 8%-10%-20% (10% concentrated ammonia in methanol solution)-dichloromethane as eluent. The product characterization data were as follows: FABMS: m/z 143.1 (MH+); HRFABMS: m/z 143.1184 (MH+), calculated value C7H15N2O: m/z 143.1184; 1H NMR (d6-DMSO) δ 1.50-1.57 (4H, m, CH2), 1.76-1.94 (4H, m. CH2), 2.10 (3H, s, -NCH3), 2.72 (1H, m, CH), 6.68-7.18 (2H, m, CONH2); 13C NMR (d6-DMSO) δ 41.2 (CH3), 28.5, 28.5, 54.9, 54.9 (CH2), 46.2 (CH), 176.7 (C).
References
[1] Journal of Medicinal Chemistry, 2003, vol. 46, # 7, p. 1116 - 1119
[2] Patent: WO2004/22561, 2004, A1. Location in patent: Page 110
[3] Journal of Medicinal Chemistry, 2005, vol. 48, # 24, p. 7520 - 7534
[4] Helvetica Chimica Acta, 1954, vol. 37, p. 1672,1676