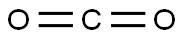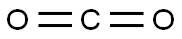Synthesis
1. 600 ml of anhydrous THF was added to a 2L reaction flask under argon protection.
2. 65.4 g of diisopropylamine was added and the reaction system was subsequently cooled to -78 °C via a dry ice acetone bath.
3. 258 ml of 2.5 M n-butyllithium solution was slowly added dropwise at 0 °C. After the dropwise addition, the reaction temperature was raised to 0 °C and maintained for 0.5 h. The reaction system was then cooled to -78 °C again.
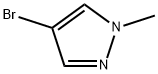
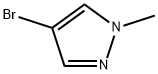
4. 80.0 g of 1-methyl-4-bromopyrazole was slowly added dropwise at -78 °C, and after the dropwise addition was completed, the reaction temperature was maintained at -78 °C for 1 hour.
5. dry carbon dioxide gas was passed into the reaction system until TLC detection showed that the reaction was complete.
6. The pH of the reaction solution was slowly adjusted to 2-3 with 1N HCl, at which time a large amount of white solid was precipitated.
7. The reaction mixture was cooled at 0 °C for 3 h, followed by filtration to collect the white solid.
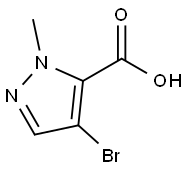
8. The resulting solid was dried at 40 °C for 16 h to give 77.3 g of white solid compound 22 (4-bromo-1-methyl-1H-pyrazole-5-carboxylic acid).