Uses
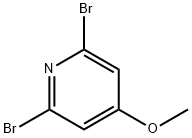
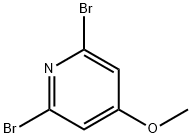
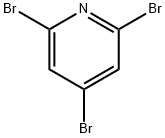
2,6-Dibromo-4-methoxypyridine is a useful reactant for the preparation of various organic compounds such as pyridine-pyridone alternate oligomers, and cyclic adenosine monophosphate.
Synthesis
Reaction of 2,4,6-tribromopyridine (5) and sodium methanol (1.2 eq.) under methanol reflux conditions afforded 2,6-dibromo-4-methoxypyridine (6) in 80% yield. Subsequently, the compound (6) was treated with n-butyllithium (1.2 eq.) at -78 °C and reacted with pivaleronitrile (1.2 eq.) for 150 min, followed by refluxing in two conventional sulfuric acids for 2 h to afford the keto isomer (7) in 86% yield. Compound (7) was converted to optically active alcohol (8) in 93% yield and 90% optical purity by hydrogen transfer type asymmetric reduction catalyzed by asymmetric ruthenium catalyst RuCl[(S,S)-Tddpen] (p-isopropylbenzene, 0.01 equiv.) using formic acid (4.3 equiv.) and triethylamine (2.5 equiv.). Next, compound (8) was converted to a camphor ester using a chloride, processed for optical splitting by recrystallization (75% yield, diastereoisomer ratio = 99/<1), and saponified again to give an almost optically pure alcohol (7, quantitatively). Finally, homogeneous coupling of compound (7) using palladium catalyst [PdCl2(PhCN)2-TDAE] afforded the pyridine isomer (9) (chemical formula 5) in 36% yield (diastereoisomer ratio = >99.5/<0.5).