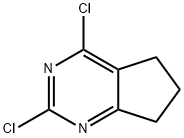
Synthesis
General procedure for the synthesis of 2,4-dichloro-5,6-trimethylpyrimidine from 6,7-dihydro-1H-cyclopenta[d]pyrimidine-2,4(3H,5H)-dione: 6,7-dihydro-1H-cyclopenta[d]pyrimidine-2,4(3H,5H)-dione (0.3 g, 1.97 mmol) was suspended in phosphorochloridic acid (5 ml) and heated to reflux overnight. Upon completion of the reaction, the reaction mixture was cooled to room temperature followed by slow addition of water under ice bath conditions. The precipitated product was collected by filtration and dried to give 2,4-dichloro-5,6-trimethylpyrimidine as a yellow solid. Yield: 92%. 1H NMR (DMSO-d6, 300 MHz): δ 3.13 (t, 2H, cyclopentyl-CH2), 3.00 (t, 2H, cyclopentyl-CH2), 2.23 (m, 2H, cyclopentyl-CH2).
References
[1] European Journal of Medicinal Chemistry, 1980, vol. 15, # 4, p. 317 - 322
[2] European Journal of Medicinal Chemistry, 2015, vol. 92, p. 246 - 256
[3] Patent: US2010/144731, 2010, A1. Location in patent: Page/Page column 22
[4] Patent: US2007/281949, 2007, A1. Location in patent: Page/Page column 93-94
[5] Patent: US2014/179668, 2014, A1. Location in patent: Paragraph 0829
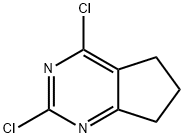

![6,7-dihydro-5H-cyclopenta[d]pyrimidine-2,4-diol](/CAS/GIF/5466-00-2.gif)