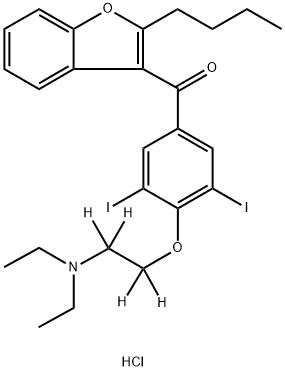
Description
Amiodarone-d
4 is intended for use as an internal standard for the quantification of amiodarone by GC- or LC-MS. Amiodarone is a class III antiarrhythmic agent, in that it prolongs both cardiac action potential and refractoriness by blocking potassium currents. It inhibits the voltage-gated potassium channel hERG, also known as KCNH2, with an IC
50 value of 1 μM. In addition, amiodarone binds with high affinity to the sigma-1 opioid receptor, 3-β-hydroxysteroid Δ
8Δ
7 isomerase, and C-8 sterol isomerase (K
is = 1, 25, and 62 nM, respectively) and inhibits human thyroid hormone receptors α and β (IC
50s = 0.6 and 0.65 μM, respectively). It also inhibits the cytochrome P450 (CYP) isoforms CYP2C8 and CYP3A4
in vitro at low micromolar concentrations.
Uses
A Labelled non-selective ion channel blocker. Antiarrhythmic (class III).
General Description
A Certified Spiking Solution
and stable-labeled internal standard for use in therapeutic drug monitoring analyses by LC/MS. Amiodarone, an antiarrhythmic agent marketed as Pacerone
, Cordarone
, Aratac, Atlansil, is monitored by clinical labs to ensure patients remain within the drug′s therapeutic range.
References
[1] T. J. CAMPBELL K. M W. Therapeutic drug monitoring: antiarrhythmic drugs[J]. British journal of clinical pharmacology, 2003, 46 4: 307-319. DOI:
10.1046/j.1365-2125.1998.t01-1-00768.x[2] NANDITA SINHA Srikanta S. Predicting hERG activities of compounds from their 3D structures: Development and evaluation of a global descriptors based QSAR model[J]. European Journal of Medicinal Chemistry, 2011, 46 2: Pages 618-630. DOI:
10.1016/j.ejmech.2010.11.042[3] CHRISTIAN LAGGNER. Discovery of High-Affinity Ligands of σ1 Receptor, ERG2, and Emopamil Binding Protein by Pharmacophore Modeling and Virtual Screening[J]. Journal of Medicinal Chemistry, 2005, 48 15: 4754-4764. DOI:
10.1021/jm049073+[4] BO CARLSSON. Synthesis and Preliminary Characterization of a Novel Antiarrhythmic Compound (KB130015) with an Improved Toxicity Profile Compared with Amiodarone[J]. Journal of Medicinal Chemistry, 2002, 45 3: 623-630. DOI:
10.1021/jm001126+[5] THOMAS M POLASEK. Mechanism-based inactivation of human cytochrome P4502C8 by drugs in vitro.[J]. Journal of Pharmacology and Experimental Therapeutics, 2004, 311 3: 996-1007. DOI:
10.1124/jpet.104.071803