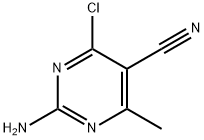
Synthesis
In a 100 mL round bottom flask, 4-chloro-6-methylpyrimidin-2-amine (3 g, 21.0 mmol) was dissolved in 40 mL of glacial acetic acid and cooled in an ice bath to less than 10C. The reaction was carried out in an ice bath. To the upper solution was added NIS (2.4 g, 10.6 mmol), the reaction was carried out for 1 hour and then NIS (2.4 g, 10.6 mmol) was added, the reaction was carried out for 1 hour and then naturally brought to room temperature for 8 hours. After the reaction was completed, the reaction solution was poured into ice water, extracted with EtOAc, the organic phase was washed with 5% Na2SO3 solution, 10% NaHCO3 solution and saturated NaCl solution, dried with anhydrous Na2SO4, filtered, and then concentrated under reduced pressure to remove the solvent, obtaining 4.9 g of solids, which was 2-amino-4-chloro-6-methylpyrimidine-5-carbonitrile, yield 87%.