Synthesis
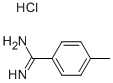
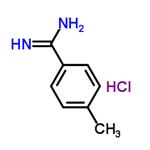
Example 7: Synthesis of 4-methylbenzamidine hydrochloride
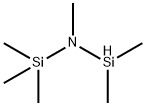
Methanol (96 mg) was added dropwise to a solution of hexachloromethyl disilazane (484 mg) in dichloromethane (1 mL) at room temperature. Subsequently, the reaction mixture was cooled in an ice bath and trimethylsilyl trifluoromethanesulfonate (667 mg) was added slowly and dropwise. The resulting suspension was stirred at room temperature for 1 h. After that, p-toluenenitrile (351 mg) was added dropwise and the stirring was continued for 48 h at room temperature. Upon completion of the reaction, the suspension was poured into 2N aqueous sodium hydroxide solution and extracted with dichloromethane. The combined organic phases were dried over anhydrous sodium sulfate, followed by evaporation under reduced pressure to remove the solvent. The crude product was purified by chromatography on an aminosilica gel column (eluent: dichloromethane/methanol = 5/1), then to the purified product was added a 4N ethyl acetate solution of hydrogen chloride, and the solvent was again removed by evaporation under reduced pressure to give the final product, 4-methylbenzamidine hydrochloride (7 mg, 2% yield).
1H-NMR (270 MHz, DMSO-d6) δ: 2.41 (3H, s), 7.43 (2H, d, J=8.0 Hz), 7.74 (2H, d, J=8.0 Hz), 9.06 (2H, s), 9.30 (2H, s).