Description
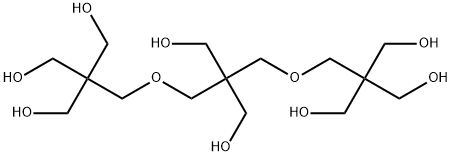
Tripentaerythritol, 2,2-bis{[hydroxy- 2,2-bis(hydroxymethyl)propoxy]methyl}-1,3- propanediol, is only slightly soluble in water and can be isolated during the production of dipentaerythritol. It has at present only a very limited economic importance. It is possibly suitable for the same applications as dipentaerythritol.
Synthesis
A method for the preparation of tripentaerythritol, comprising the basic steps of condensation,distillation and deformalization, evaporation and crystallization, centrifugation, drying, etc., characterized in that it also comprises the following steps: firstly, 50.4m3 of formaldehyde with the concentration of 31%, 5.25m3 of acetaldehyde with the concentration of 98%, and 9.45m3 of liquid alkali with the concentration of 2.88% are prepared in the high-level tank respectively, and then 31% formaldehyde is put into the condensation tubular reactor at the speed of 16.8m3/h. Then the concentration of 31% formaldehyde was put into the condensation tubular reactor at the rate of 16.8m3/h. After 2 minutes of formaldehyde feeding, liquid alkali was put into the condensation tubular reactor at the rate of 3.15m3/h. Finally, after 2 minutes of liquid alkali feeding, acetaldehyde was put into the condensation tubular reactor at the rate of 1.75m3/h. When the temperature of the tubular reactor reaches 45, the cooling water was turned on to control the rate of heating up, and the rate of heating up was 1/5min until all the feeding was completed. The feeding is completed, the feeding of the completed reaction solution is transferred to the three condensation kettle, stirring until the reaction is complete, after holding for 10 minutes, with a concentration of 85% of the formic acid to neutralize the solution PH value of 6 ~ 6.5, when the temperature is 60 , the condensed solution containing three quaternary amyl alcohol totaling 75m3 is sent to the settling tank, open the stirring, stirring speed of 15r/min, and circulating water to cool down to 25 , then holding for two two hours, the cooling rate is at 2 hours. The cooling rate was 2C/h. After the condensate was settled for 24 hours, the condensate was sent to a filter press for filtration, so that the moisture content in the filter cake was less than 20%. The filtered cake was pressed to obtain 200 kilograms of wet material, which was sent to a rotary kiln dryer for the first time drying to a moisture content of 5%, and further dried in a rotary dryer to a moisture content of the cake of less than 1%, to obtain 140 kilograms of finished product tritiated amyl alcohol.