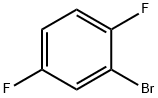
Chemical Properties
CLEAR COLOURLESS LIQUID
Uses
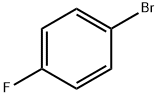
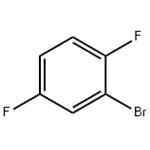
2-Bromo-1,4-difluorobenzene has been used in the preparation of 1,4-difluoroanthraquinone, a precursor to ametantrone.
General Description
Lithiation of 2-bromo-1,4-difluorobenzene in the presence of lithium diisopropylamide (LDA) in THF-hexane, butyllithium in diethyl ether-hexane and butyllithium in THF-hexane has been reported.
Synthesis
GENERAL STEPS: In a FEP or PFA reactor equipped with a PTFE-lined magnetic stir bar and connected to a gas scrubber bottle, substituted benzene (0.95-1.10 mmol), 1,1,1,3,3-pentafluorobutane (2 mL per mL of C6H5R), and BF3-Et2O (1.3-1.5 mmol/mmol C6H5R) were added. The mixture was stirred at 0-5 °C (ice bath) for 10-15 min, followed by batchwise addition of XeF2 (1.2-1.3 mmol/mmol C6H5R). After each addition, the mixture was stirred at 22-25 °C for 3-5 min and cooled to 0-5 °C again. After the addition was completed, the dark-colored solution was continued to be stirred at 22-25 °C for 15-30 min. Upon completion of the reaction, the reaction was quenched by the addition of 10% aqueous KHCO3, the upper organic layer was separated and filtered through a short column packed with silica gel (40-60 μm) and finally dried with magnesium sulfate. The solution was analyzed by 19F NMR and GC/MS. The major products are listed in the table and the others are as follows (GC/MS data provided).
References
[1] Russian Journal of Organic Chemistry, 2016, vol. 52, # 10, p. 1400 - 1407
[2] Zh. Org. Khim., 2016, vol. 52, # 10, p. 1412 - 1419,8