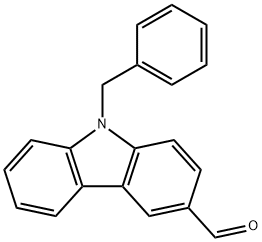
Synthesis
9-Benzylcarbazole-3-carbaldehyde was prepared as follows:
1) 1:1 equivalent of carbazole with benzyl bromide, DMF as solvent, add 5 equivalent of potassium carbonate, room temperature reaction 8h. After the reaction, recrystallized with anhydrous ethanol, to obtain a white solid carbazole-3-carbaldehyde, yield 70-85%.
(2) 0 ice bath, equal volume of N, N-dimethylformamide and trichlorophosphorus reaction 30min, add the compound dissolved in dichloromethane carbazole-3-carboxaldehyde, 90 reaction 6h. After the end of the reaction, quenched with water, sodium hydroxide and neutralized to pH = 7, with ethyl acetate and water extraction, the organic phase was collected, and the silica gel column purification yielded a white solid 9-benzylcarbazole-3-carboxaldehyde, the yield was 7789%. 89%.