Synthesis
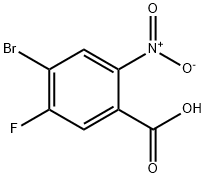
General procedure for the synthesis of 2-amino-4-bromo-5-fluorobenzoic acid from 4-bromo-5-fluoro-2-nitrobenzoic acid: 5% Pt/C (50% aqueous wet, 60 g, 6 wt%) was added to a nitrogen-protected reaction vessel containing isopropyl acetate (22 L) and 4-bromo-5-fluoro-2-nitrobenzoic acid (1.00 kg, 3.79 mol). The headspace of the reaction vessel was displaced three times with nitrogen and then three times with hydrogen. The reaction mixture was stirred at 25 °C and under hydrogen atmosphere. after 40 h, the reaction was completed and the headspace was again replaced three times with nitrogen. The reaction mixture was filtered and the reaction vessel and filter were rinsed with isopropyl acetate (5 L). The organic layers were combined and concentrated under reduced pressure to 5.0 L. Subsequently, the solvent was replaced with toluene under reduced pressure, and the solid obtained was separated by filtration, washed with toluene, and dried under reduced pressure at 50 °C to give 0.59 kg (66% yield) of 2-amino-4-bromo-5-fluorobenzoic acid as a white to off-white crystalline solid.
References
[1] Organic Letters, 2018, vol. 20, # 13, p. 3736 - 3740
[2] Patent: WO2018/118830, 2018, A1. Location in patent: Page/Page column 25-26
[3] Patent: WO2017/192304, 2017, A1. Location in patent: Paragraph 0378