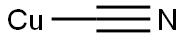Synthesis
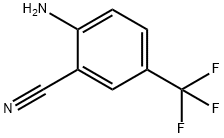
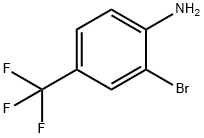
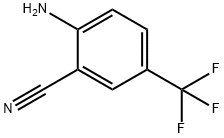
A reaction was carried out with cuprous cyanide (2.24 g, 25.00 mmol) and 2-bromo-4-(trifluoromethyl)aniline (5.0 g, 20.83 mmol) in N-methyl-2-pyrrolidone (20 mL) heated at 195 °C for 4 hours. After completion of the reaction, the reaction mixture was diluted with ethyl acetate (100 mL) and washed twice with 28% aqueous ammonium hydroxide solution, twice with saturated aqueous sodium chloride solution (brine), and twice with water, in that order. The organic layer was separated, dried over anhydrous sodium sulfate and concentrated under reduced pressure to remove the solvent. The resulting residue was purified by fast chromatography using hexane as the starting eluent and gradually increasing the proportion of ethyl acetate to a gradient elution of 30% hexane-70% ethyl acetate, resulting in 2-amino-5-trifluoromethylbenzonitrile (1.821 g, 9.78 mmol, 47% yield) as a green amorphous solid. Mass spectrum (m/e): 187 (M + 1).