Synthesis
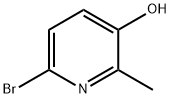
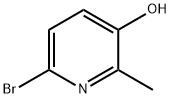
Step 2: Synthesis of 6-bromo-2-methylpyridin-3-ol
4,6-Dibromo-2-methylpyridin-3-ol (15.8 g, 59.4 mmol) was dissolved in tetrahydrofuran (THF, 200 mL). The reaction solution was cooled to -78°C and n-butyllithium (n-BuLi, 50 mL, 125 mmol, 2.5 M hexane solution) was slowly added dropwise at this temperature. After the dropwise addition, the reaction temperature was maintained at -78°C and stirring was continued for 2 hours. Upon completion of the reaction, the reaction was quenched with deionized water (50 mL) and the reaction solution was neutralized with 2 N hydrochloric acid (HCl). Subsequently, the aqueous phase was extracted with dichloromethane (2 x 50 mL). The organic phases were combined, dried with anhydrous sodium sulfate (Na2SO4) and concentrated under reduced pressure to afford 6-bromo-2-methylpyridin-3-ol (10.5 g, 95% yield) as a yellow oil. The product was confirmed by nuclear magnetic resonance hydrogen spectroscopy (1H NMR, 300 MHz, DMSO-d6): δ 2.29 (s, 3H), 7.08 (d, 1H), 7.26 (d, 1H), 10.08 (s, 1H).
References
[1] Patent: WO2012/112743, 2012, A1. Location in patent: Page/Page column 153-154
[2] Patent: WO2012/125613, 2012, A1. Location in patent: Page/Page column 139
[3] Patent: US2012/196869, 2012, A1. Location in patent: Paragraph 0338; 0339
[4] Patent: WO2013/109521, 2013, A1. Location in patent: Paragraph 00261; 00262
[5] Synlett, 2003, # 11, p. 1678 - 1682