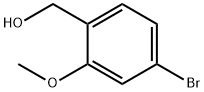
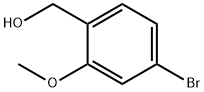
Chemical Properties
Colorless to light yellow fine crystalline powder
Uses
It is used in azaquinone-methide-mediated depolymerization of aromatic carbamate oligomers.
Synthesis
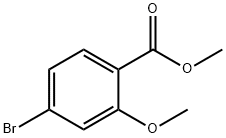
General procedure for the synthesis of 4-bromo-2-methoxybenzenemethanol from methyl 4-bromo-2-methoxybenzoate: To a solution of methyl 4-bromo-2-methoxybenzoate (7.14 g, 29 mmol) in tetrahydrofuran (THF, 35 mL) was slowly added a THF solution of 2 M lithium borohydride (LiBH4) (35 mL, 70 mmol, 2.4 equiv). The reaction mixture was heated to reflux at 60 °C for 16 hours. Upon completion of the reaction, the mixture was cooled to room temperature and the solvent was evaporated under reduced pressure. The residue was dissolved in 5% aqueous acetic acid (pH adjusted to 6) and stirred for 15 minutes. Subsequently, the aqueous layer was extracted with ethyl acetate (EtOAc, 2 x 50 mL), the organic phases were combined and washed with saturated brine, dried over anhydrous magnesium sulfate (MgSO4), filtered and concentrated to give the crude product as a tan oil. The crude product was purified by fast column chromatography using 5% ethyl acetate in hexane solution as eluent to give 5.96 g (94% yield) of the target product 4-bromo-2-methoxybenzenemethanol as a white solid: Thin Layer Chromatography (TLC) Rf value 0.30 (10% ethyl acetate/hexane); 1H NMR (CDCl3) δ 7.30 (d, 1H), 7.14 (d, 1H), 7.11 (d, 1H). 1H), 7.11 (s, 1H), 5.09 (t, 1H), 4.42 (d, 2H), 3.77 (s, 3H).
References
[1] Bioorganic and Medicinal Chemistry, 2014, vol. 22, # 1, p. 419 - 434
[2] Journal of Medicinal Chemistry, 1992, vol. 35, # 4, p. 734 - 740
[3] Patent: WO2006/60461, 2006, A1. Location in patent: Page/Page column 197
[4] ChemMedChem, 2010, vol. 5, # 1, p. 65 - 78
[5] Patent: WO2012/66335, 2012, A1. Location in patent: Page/Page column 54