Synthesis
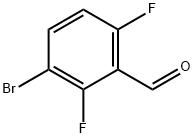
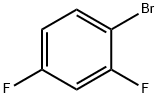
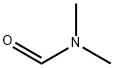
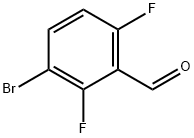
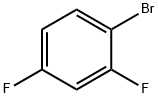
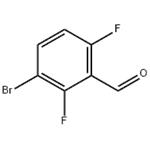
Lithium diisopropylammonium (2M THF solution, 13 mL, 26 mmol) was slowly added dropwise to a solution of anhydrous THF (80 mL) of 1-bromo-2,4-difluorobenzene (5.0 g, 25.9 mmol) at -78 °C, and the reaction was stirred for 1 h at -78 °C maintaining the temperature. Subsequently, N,N-dimethylformamide (2.0 g, 28 mmol) was added to the reaction system and stirring was continued at -78 °C for 30 min. After completion of the reaction, the reaction was quenched by adding water to the reaction mixture and extracted with ethyl acetate. The organic phases were combined, washed sequentially with 1N hydrochloric acid and water, dried over anhydrous magnesium sulfate, and concentrated under reduced pressure to remove the solvent. The residue was purified by silica gel column chromatography (eluent: ethyl acetate/hexane=1:9) to give 3.0 g of 3-bromo-2,6-difluorobenzaldehyde in 32% yield. The melting point of the product was 53-54 °C. 1H-NMR (CDCl3) δ: 7.00 (1H, dt, J = 9.3,1.8 Hz), 7.71-7.82 (1H, m), 10.3 (1H, s).
References
[1] Patent: US2010/41891, 2010, A1. Location in patent: Page/Page column 62
[2] Patent: WO2015/89809, 2015, A1. Location in patent: Page/Page column 49; 50
[3] Patent: WO2015/95256, 2015, A1. Location in patent: Page/Page column 50