Synthesis
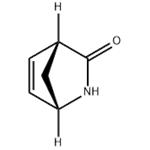
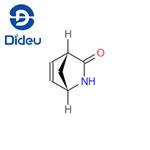
GENERAL METHOD: Unreacted N-hydroxymethyl-γ-lactams (1S,4R)-1 and (1R,4S)-2 (20 mg, 0.14 mmol) were dissolved in methanol (2 mL). Subsequently, ammonium hydroxide (2 mL) was added and the reaction mixture was stirred at room temperature for 4 hours. Upon completion of the reaction, the solvent was removed by rotary evaporator and the residue was purified by silica gel column chromatography, using ethyl acetate as eluent, to afford white crystals of (1S,4R)-3 [15 mg, 95%; [α]D25 = +545 (c 0.3, CHCl3), ee = 99%; literature value 13: [α]D25 = +549 (c 0.26, CHCl3), ee > 99%; melting point 95-98°C (recrystallized from i-Pr2O), literature value 13: 97-100°C] or (1R,4S)-4 [15 mg, 93%; [α]D25 = +125.5 (c 0.55, CHCl3), ee = 96%; literature value 13: [α]D25 = +158 (c 0.45, CHCl3), ee > 99%; melting point 78-81°C (recrystallized from i-Pr2O), literature value 13: 78-81°C]. (1H NMR (400 MHz, CDCl3, 25 °C, TMS) data for (1S,4R)-3: δ = 2.18-2.43 (m, 2H, CH2); 3.18-3.24 (m, 1H, CHCO); 4.30-4.37 (m, 1H, CHNH); 6.05 (br s, 1H, NH); 6.63-6.82 ( m, 2H, CH=CH). Elemental analysis: calculated values (C6H7NO): C, 66.04; H, 6.47; N, 12.84; measured values: C, 66.12; H, 6.38; N, 12.82. 1H NMR (400 MHz, CDCl3, 25 °C, TMS) data for (1R,4S)-4: δ = 1.33-1.97 (m, 6H, 3 × CH2) ; 2.76-2.85 (m, 1H, CHCO); 3.04 (br s, 1H, NH); 3.95-4.08 (m, 1H, CHNH). Elemental analysis: calculated values (C6H9NO): C, 64.84; H, 8.16; N, 12.60; measured values: C, 64.81; H, 8.13; N, 12.68.
References
[1] Tetrahedron Asymmetry, 2016, vol. 27, # 16, p. 729 - 731
[2] Tetrahedron Asymmetry, 1996, vol. 7, # 8, p. 2381 - 2386
[3] Tetrahedron: Asymmetry, 1994, vol. 5, # 7, p. 1155 - 1156
![(1S,4R)-2-Aza-bicyclo[2.2.1]hept-5-en-3-one Structure](https://www.chemicalbook.com/CAS/GIF/130931-83-8.gif)

![2-Azabicyclo[2.2.1]hept-5-en-3-one, 2-(hydroxymethyl)-, (1S,4R)-](/CAS/20200331/GIF/157810-20-3.gif)
![2-Azabicyclo[2.2.1]hept-5-en-3-one](/CAS/GIF/49805-30-3.gif)