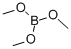Synthesis
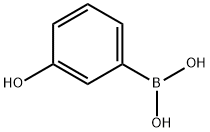
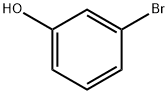
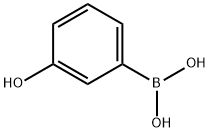
(vi) Preparation of 3-hydroxyphenylboronic acid: m-Bromophenol (8.65 g, 50 mmol) was dissolved in anhydrous tetrahydrofuran (150 mL) and sodium hydride (60%, 2.4 g, 60 mmol) was added at room temperature. After 1 hour of reaction, sec-butyl lithium (1.3 M, 50 mL, 65 mmol) was added slowly and dropwise to the reaction solution at -78 °C. Stirring was continued at this temperature for 30 min, followed by the addition of trimethyl borate (15 mL). The reaction system was gradually warmed to room temperature and maintained for 2 hours. Upon completion of the reaction, the reaction was quenched with deionized water (50 mL) and extracted with dichloromethane (2 x 100 mL). The organic phases were combined, dried with anhydrous magnesium sulfate and concentrated under reduced pressure to afford 3-hydroxyphenylboronic acid as a white solid (4.0 g, 58% yield), and the product was used directly in the subsequent Suzuki coupling reaction without further purification.