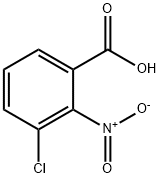
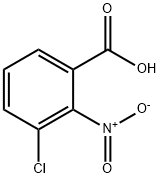
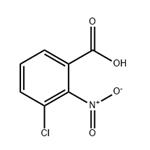
Chemical Properties
almost white to beige crystalline powder
Uses
3-Chloro-2-nitrobenzoic acid has been used in the preparation of:
- 2-amino-3-chlorobenzonitrile
- 3-chloro-2-nitrobenzaldehyde
General Description
Hydrogen-bonded structures of isomeric compounds of 3-chloro-2-nitrobenzoic acid with quinoline have been investigated.
Synthesis
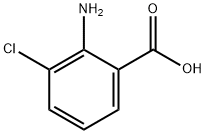
General procedure for the synthesis of 3-chloro-2-nitrobenzoic acid from 2-amino-3-chlorobenzoic acid: 216 g of glacial acetic acid was added to a 500 ml triple-necked flask, a condensing unit was installed and stirring was turned on. Subsequently, 57 g of sodium borate and 5 g of titanate catalyst prepared in Example 1 were added. The internal air of the system was replaced with nitrogen three times and the reaction temperature was raised to 85°C. Under stirring conditions, a mixed solution comprising 10 g of 2-amino-3-chlorobenzoic acid and 30 g of glacial acetic acid was slowly added dropwise. The reaction temperature was maintained at 85°C-90°C and the reaction was stirred for 1 hour. The reaction progress was monitored by thin layer chromatography (TLC). After completion of the reaction, the system was cooled to room temperature and the reaction mixture was filtered. The resulting solid was washed with 20 g of methanol and subsequently dried under vacuum at 40°C -50°C for 3 h to give 11.3 g of 2-nitro-3-chlorobenzoic acid crude product. 40 g of the crude product was dissolved in anhydrous ethanol, 2-nitro-3-chlorobenzoic acid was added and heated to 75°C -80°C refluxing for 0.5 hours. The temperature of the reaction solution was then lowered to 0°C -5°C, held and stirred for 0.5 hours and filtered. The solid was dried under vacuum at 40°C -50°C for 3 hours to obtain 10 g of 2-nitro-3-chlorobenzoic acid with 99.7% purity and 84.9% yield.
References
[1] Patent: CN104086429, 2016, B. Location in patent: Paragraph 0040; 0041; 0042