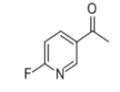
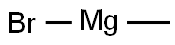Synthesis
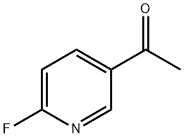
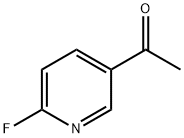
To a cold solution (-78°C) of 6-fluoro-N-methoxy-N-methylnicotinamide (Method 38, 7.2 g, 39 mmol) in tetrahydrofuran (130 mL) was added slowly and dropwise methylmagnesium bromide (20 mL, 59 mmol, 3 M ether solution). The cooling bath was removed and the reaction mixture was allowed to gradually warm to room temperature with continuous stirring for 2 hours. Upon completion of the reaction, the reaction was quenched with 3 N hydrochloric acid solution and the layers were separated. The organic layer was dried over anhydrous sodium sulfate, filtered and concentrated under reduced pressure to give 3.8 g of 2-fluoro-5-acetylpyridine in 70% yield. The product was characterized by 1H NMR (CDCl3): δ 8.80 (s, 1H), 8.29-8.43 (m, 1H), 6.98-7.06 (m, 1H), 2.62 (s, 3H).
References
[1] Patent: WO2006/82392, 2006, A1. Location in patent: Page/Page column 116
[2] Patent: US2017/313683, 2017, A1. Location in patent: Paragraph 1251-1252