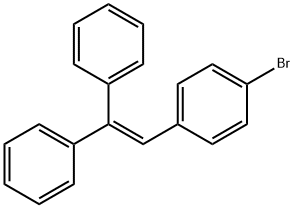
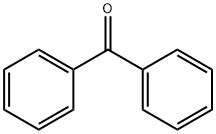
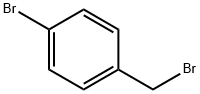
Synthesis
General procedure for the synthesis of 2-(4-bromophenyl)-1,1-diphenylethylene from benzophenone and p-bromobenzyl bromide: 40.00 g of p-bromobenzyl bromide (0.16 mol) and 83.6 mL of triethyl phosphite (0.48 mol) were added to a 500 mL three-necked flask, and the reaction was heated under argon protection in an oil bath at 150 °C for 24 h with magnetic stirring, and then the reaction was The reaction was cooled to room temperature to obtain the Yerid reagent. To another three-necked flask was added 450 mL of tetrahydrofuran (THF) followed by 21.87 g of benzophenone (0.12 mol) and 40.39 g of potassium tert-butanolate (t-BuOK, 0.36 mol). The mixture was stirred for 24 hours. Upon termination of the reaction, the reaction solution was poured into water, which produced a large white precipitate, which was collected by diafiltration. The precipitate was washed three times with ethanol and dried under vacuum at 60 °C for 24 h to give the white intermediate Tri-Br.
References
[1] Patent: CN104341311, 2016, B. Location in patent: Paragraph 0028; 0029; 0030
[2] Chinese Journal of Chemistry, 2015, vol. 33, # 8, p. 939 - 947