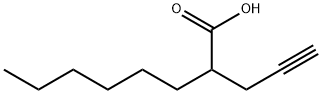
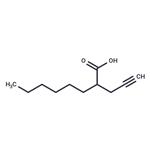
Description
2-
hexyl-
4-
Pentynoic acid is a derivative of valproic acid , an inhibitor of histone deacetylases (HDACs). It inhibits HDAC activity more potently (IC
50 = 13 μM) than valproic acid (IC
50 = 398 μM). 2-
hexyl-
4-
Pentynoic acid induces histone hyperacetylation in cerebellar granule cells significantly at 5 μM. It also induces the expression of heat shock proteins Hsp70-
1a and Hsp70-
1b and protects cerebellar granule cells from glutamate-
induced excitotoxicity when used at a concentration of 50 μM.
Uses
2-Hexyl-4-pentynoic acid ((±)-2-Hexyl-4-pentynoic acid), valproic acid (VPA) derivative, exhibits potential roles of HDAC inhibition (IC50=13 μM) and HSP70 induction. Potent neuroprotective effects. 2-Hexyl-4-pentynoic acid causes histone hyperacetylation and protect against glutamate-induced excitotoxicity in cultured neurons[1]. 2-Hexyl-4-pentynoic acid is a click chemistry reagent, it contains an Alkyne group and can undergo copper-catalyzed azide-alkyne cycloaddition (CuAAc) with molecules containing Azide groups.
References
[1] DANIEL EIKEL Heinz N Alfonso Lampen. Teratogenic Effects Mediated by Inhibition of Histone Deacetylases: Evidence from Quantitative Structure Activity Relationships of 20 Valproic Acid Derivatives[J]. Chemical Research in Toxicology, 2006, 19 2: 272-278. DOI:
10.1021/tx0502241[2] YAN LENG . Potent neuroprotective effects of novel structural derivatives of valproic acid: Potential roles of HDAC inhibition and HSP70 induction[J]. Neuroscience Letters, 2010, 476 3: Pages 127-132. DOI:
10.1016/j.neulet.2010.04.013