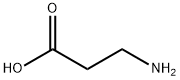
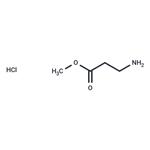
Synthesis
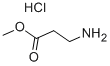
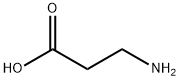
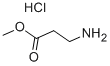
Under ice bath conditions, 60 mL of methanol was added to a 100 mL round-bottom flask, followed by the slow dropwise addition of 4 mL of sulfoxide chloride (SOCl2) through a constant-pressure dropping funnel (with a desiccant tube at the top), and the resulting off-gas was absorbed with NaOH solution. After stirring the reaction mixture for 1 h, 8 mmol of β-alanine was added and stirring was continued for 30 min at room temperature. Subsequently, the reaction system was warmed up to 66 °C and refluxed for 6 hours. The reaction process was monitored by thin-layer chromatography (TLC) using a 2% ethanol solution of ninhydrin as a color developer until the raw material spot disappeared. Upon completion of the reaction, the solvent was removed by distillation to afford methyl 3-aminopropionate hydrochloride in 100% yield.
References
[1] European Journal of Organic Chemistry, 2012, # 29, p. 5774 - 5788,15
[2] European Journal of Organic Chemistry, 2012, # 29, p. 5774 - 5788
[3] Patent: US2014/206741, 2014, A1. Location in patent: Paragraph 0016
[4] European Journal of Medicinal Chemistry, 2018, vol. 154, p. 44 - 59
[5] Journal of Organic Chemistry, 1991, vol. 56, # 7, p. 2553 - 2557