Synthesis
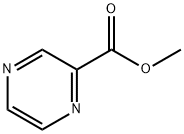
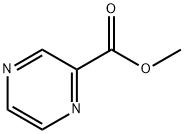
To a solution of 2-pyrazinecarboxylic acid (5.80 g, 46.5 mmol) in dichloromethane (100 mL) was slowly added oxalyl chloride (5.10 mL, 60.4 mmol), followed by the addition of a catalytic amount of DMF.The reaction mixture was stirred for 6 hr. at 23° C. The reaction was completed with the addition of methanol (30 mL). After completion of the reaction, methanol (30 mL) was added slowly and stirring was continued for 30 min. The reaction mixture was concentrated under reduced pressure. The residue was dissolved in ethyl acetate (200 mL) and washed sequentially with saturated aqueous sodium bicarbonate (1 x 100 mL) and brine (1 x 100 mL). The organic layer was dried with anhydrous magnesium sulfate, filtered and concentrated under reduced pressure to afford 3.38 g (53% yield) of the crude methyl pyrazine-2-carboxylate, which could be used in the next step of the reaction without further purification.1H NMR (400 MHz, CDCl3): δ 9.34 (d, 1H), 8.79 (d, 1H), 8.74 (dd, 1H), 4.06 (s 3H); LCMS (Method A): tR = 0.48 min, m/z 139.3 (M + H)+.
References
[1] Bioorganic and Medicinal Chemistry, 2016, vol. 24, # 12, p. 2843 - 2851
[2] Journal of the American Chemical Society, 1990, vol. 112, # 17, p. 6248 - 6254
[3] Patent: WO2004/48369, 2004, A1. Location in patent: Page 8
[4] Tetrahedron Letters, 2009, vol. 50, # 28, p. 4030 - 4032
[5] Patent: WO2016/207914, 2016, A2. Location in patent: Page/Page column 3; 13; 14