Synthesis
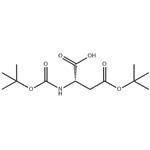
2.1. Preparation of Boc-Asp(O-tBu)-NCA (II-a)
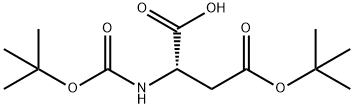
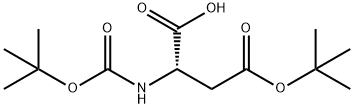
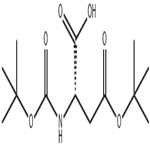
The method involves the main chain cyclization of a free carboxylic acid in which the amine functional group is protected by Boc. First, (Boc)2-Asp(Ot-Bu)-OH (VII-a) needs to be prepared. The specific steps are as follows:
1. esterify the α-carboxylic acid of Boc-Asp(Ot-Bu)-OH (Va) to benzyl ester (VI-a).
2. reaction with Boc2O in the presence of DMAP to give the bis-Boc-protected amino compound (VII-a).
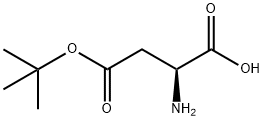
3. removal of the benzyl ester protecting group by hydrogenation to give (VIII-a).
Subsequent steps include cyclization with Vilsmeier salt protected amino acid (VIII-a). The optimum reaction conditions were in acetonitrile with the salt formed from DMF and oxalyl chloride. The final compound (II-a) was obtained in 90% yield.
References
[1] Patent: US2010/16631, 2010, A1. Location in patent: Page/Page column 5-6
[2] Bioorganic and Medicinal Chemistry Letters, 2012, vol. 22, # 15, p. 4975 - 4978
[3] Patent: WO2017/24009, 2017, A1. Location in patent: Paragraph 0652
[4] Russian Journal of Bioorganic Chemistry, 1999, vol. 25, # 5, p. 283 - 287
[5] Bioorganicheskaya Khimiya, 1999, vol. 25, # 5, p. 323 - 328