Preparation
STEP A:
Preparation of 7-chloro-3-hydrazono-oxindole.
A solution of 25.0 g of 7-chloro-isatin in 250 ml of
ethanol is refluxed for 24 hours, cooled, and precipitate
recovered by filtering and washed once with ethanol
and twice with pentane to obtain 7-chloro-3-hydrazono
oxindole, m.p. 217-219℃. (decomp.). STEP B:
Preparation of 7-chloro-oxindole.
To a solution of sodium ethoxide prepared by heating
5.5 g of sodium in 200 ml. of ethanol at 70°℃. is added
17 g of 7-chloro-3-hydrazono-oxindole over a period of
3.5 hours, and the resulting solution is heated at 70℃.
for 24 hours. The solvent is evaporated in vacuo, the
residue dissolved in water, acidified with 6 N-hydro
chloric acid and the resulting precipitate recovered by
filtering, washed three times with water, dried by suc
tion and crystallized from methylene chloride/ether to
obtain 7-chlorooxindole, 28-220℃.
Synthesis
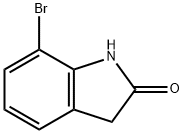
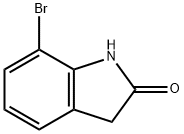
GENERAL PROCEDURE: Titanium tetrachloride (0.7 mL, 6 mmol) was slowly added to a stirred suspension of zinc powder (0.78 g, 12 mmol) in freshly distilled anhydrous tetrahydrofuran (15 mL) at room temperature and under a dry nitrogen atmosphere. After addition, the mixture was refluxed for 2 hours. After the formed suspension of low-valent titanium reagent was cooled to room temperature, a solution of 7-bromoindigo red (2 mmol) in tetrahydrofuran (10 mL) was added dropwise. The reaction mixture was stirred at room temperature for about 5 min under nitrogen protection. After confirming the completion of the reaction by thin layer chromatography (TLC) analysis, the reaction was quenched with 3% hydrochloric acid (15 mL) and subsequently extracted with chloroform (3 x 50 mL). The combined organic phases were washed with water (3 x 50 mL) and dried over anhydrous sodium sulfate. After concentration of the solvent under reduced pressure, the crude product was purified by column chromatography (petroleum ether/ethyl acetate = 5:1) to give pure 7-bromoindolone.
References
[1] Tetrahedron Letters, 2014, vol. 55, # 14, p. 2238 - 2242
[2] Tetrahedron Letters, 2000, vol. 41, # 47, p. 9089 - 9093
[3] Journal of Chemical Research, 2017, vol. 41, # 9, p. 537 - 540
[4] Patent: WO2017/199265, 2017, A1. Location in patent: Paragraph 000170