Description
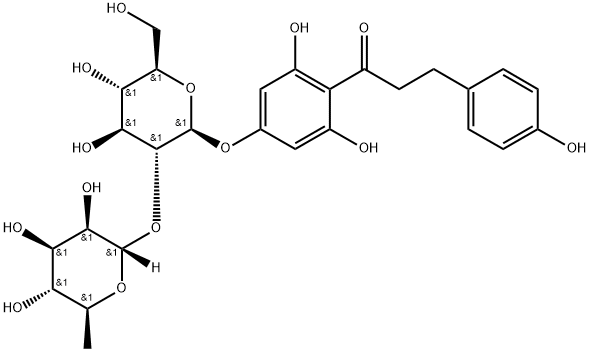
Naringin dihydrochalcone is an artificial sweetener and flavonoid that has been isolated from various plants and has antioxidant activity. It scavenges ABTS (Item No. 27317), oxygen, and 2,2-diphenyl-1-picrylhydrazyl (DPPH; Item No. 14805) radicals in cell free assays (IC50s = 24, 322.8, and 318.9 μM, respectively).
Uses
Naringin Dihydrochalcone is an intense sweetener and strong antioxidant compound which displays anti-allegeric activity.
Definition
ChEBI: Naringin dihydrochalcone is a member of flavonoids and a glycoside.
Synthesis
Naringin was used as a raw material to synthesize 1-(4-((2S,3R,4S,5S,6R)-4,5-dihydroxy-6-(hydroxymethyl)-3-(((2S,3R,4R,5R,6S)-3,4,5-trihydroxy-6-methyltetrahydro-2H-pyran-2-yl)oxy)tetrahydro-2H-pyran-2-yl)oxy)-2,6-dihydroxyphenyl)-3-( 4-hydroxyphenyl)propan-1-one was prepared in the following general procedure: 120 g of the natural extract (containing 600 g of 8% naringin) was dissolved in potassium hydroxide solution and filtered to obtain a clarified solution. The filtrate was transferred to a 2000 ml autoclave and 200 g of ethanol and 3 g of palladium carbon catalyst were added. Hydrogen was introduced at 55°C, maintaining the pressure at 1~2MPa, and the reaction was carried out for 5~7 hours until the hydrogen absorption was basically completed. The reaction solution was removed and filtered to remove the palladium-carbon catalyst. Subsequently, the hydrogenated solution was acidified with hydrochloric acid, the pH was adjusted to 5-7, and stirred thoroughly to induce crystallization, and a white solid was rapidly precipitated. The mixture was cooled to 20 °C and filtered, and the resulting wet product was dried under reduced pressure at 60 °C to give 118 g of dihydronaringenin chalcone as a white powder with 98.9% liquid phase purity and 98% molar yield. The product needs to be stored in a cool place under light nitrogen protection.
References
[1] Patent: CN105801636, 2016, A. Location in patent: Paragraph 0022
[2] Journal of Agricultural and Food Chemistry, 2003, vol. 51, # 11, p. 3309 - 3312