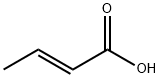
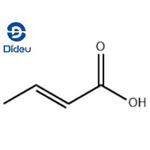
Chemical Properties
Crotonic acid is a white to light yellow crystalline flakes with a pungent odor. It is moderately soluble in water but highly soluble in organic solvents such as ethanol and ether.
Uses
trans-Crotonic acid is used in the copolymerization of crotonic acid hydrogel systems by using gamma-rays. The crosslinked copolymeric crotonic acid hydrogels release fertilizers and drugs that help to prevent environmental pollution.
Definition
ChEBI: Crotonic acid is a but-2-enoic acid with a trans- double bond at C-2. It has been isolated from Daucus carota. It has a role as a plant metabolite. It is a conjugate acid of a crotonate.
Preparation
(E)-2-Butenoic acid may be formed by oxidation of crotonaldehyde, 2-butenal. If (Z)-butenoic acid is heated at 180°C, it
is converted to (E)-2-butenoid acid.
Application
Crotonic acid is an unsaturated fatty acid containing a double bond and a carboxyl group in its molecular structure, rendering it highly reactive. It serves various industrial applications, primarily in the preparation of resins, fungicides, surface coatings, plasticisers, and pharmaceuticals.
General Description
Crotonic acid undergoes esterification with butan-2-ol by following second order reaction kinetics.
Flammability and Explosibility
Not classified
Purification Methods
Distil the acid under reduced pressure and/or recrystallise it from pet ether (b 60-80o) or water, or by partial freezing of the melt. [Beilstein 2 IV 1498.]
References
[1] VAHIDEH ELHAMI Mark A H, BOELO SCHUUR* Crotonic Acid Production by Pyrolysis and Vapor Fractionation of Mixed Microbial Culture-Based Poly(3-hydroxybutyrate-co-3-hydroxyvalerate)[J]. Industrial & Engineering Chemistry Research, 2023, 62 2: 916-923. DOI:
10.1021/acs.iecr.2c03791.
[2] ZHIQUN YU*, JIADI ZHOU Continuous Flow Synthesis of Crotonic Acid from Crotonaldehyde[J]. Organic Process Research & Development, 2024, 28 5: 1773-1782. DOI:
10.1021/acs.oprd.3c00376.
[3] KANG S, CHEN R, FU J, et al. Catalyst-free valorization of poly-3-hydroxybutyrate to crotonic acid†[J]. Reaction Chemistry & Engineering, 2021, 10: 1791-1795. DOI:
10.1039/D1RE00273B.