Synthesis
The general procedure for the synthesis of 1,3,4,5-tetrahydro-benzo[b][1,4]diazepin-2-one from the compound (CAS: 1270914-58-3) is as follows:
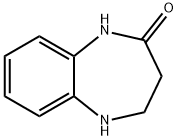
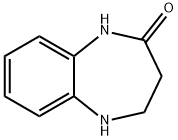
Step 3: Synthesis of 4,5-dihydro-1H-benzo[b][1,4]diazepin-2(3H)-one
1. methanol (1.8 g, 77.2 mmol) was added in batches to methanol (50 mL) at 0 °C and the mixture was stirred for 1 hour.
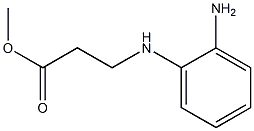
2. the above mixture was added dropwise to a solution of methyl 3-((2-aminophenyl)amino)propionate (9.4 g, 48.2 mmol) in methanol (150 mL), keeping the temperature at 0 °C. the mixture was then stirred for 1 h. The mixture was then added to the methanol (50 mL) by dropwise addition.
3. After completion of the dropwise addition, the reaction mixture was stirred at room temperature overnight.
4. The reaction mixture was concentrated and the residue was purified by CombiFlash fast column chromatography (eluent: petroleum ether/ethyl acetate = 5:1) to afford 4,5-dihydro-1H-benzo[b][1,4]diazepin-2(3H)-one as a yellow solid (3.9 g, 50% yield).
Product Characterization:
- LRMS (M + H)+: m/z 161
- 1H NMR (300 MHz, CD3OD): δ 2.61 (t, J = 6.0 Hz, 2H), 3.62 (t, J = 6.0 Hz, 2H), 6.76-6.86 (m, 2H), 6.90-6.99 (m, 2H).
References
[1] Patent: WO2012/151512, 2012, A2. Location in patent: Page/Page column 113-114
[2] ACS Medicinal Chemistry Letters, 2016, vol. 7, # 2, p. 145 - 150