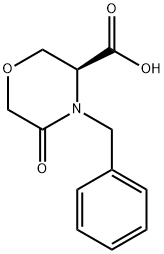
(3S)-4-Benzyl-5-oxomorpholine-3-carboxylic acid
- Product Name(3S)-4-Benzyl-5-oxomorpholine-3-carboxylic acid
- CAS106973-37-9
- CBNumberCB61512397
- MFC12H13NO4
- MW235.24
- MDL NumberMFCD09835535
- MOL File106973-37-9.mol
- MSDS FileSDS
Chemical Properties
| storage temp. | Sealed in dry,Room Temperature |
(3S)-4-Benzyl-5-oxomorpholine-3-carboxylic acid Price
| Product number | Packaging | Price | Product description | Buy |
|---|---|---|---|---|
| TRC B194183 | 500mg | $110 | (S)-4-Benzyl-5-oxomorpholine-3-carboxylicAcid |
Buy |
| Activate Scientific AS21712 | 1g | $119 | (S)-4-Benzyl-5-oxomorpholine-3-carboxylic acid 97+% ee |
Buy |
| AK Scientific 7415CU | 10g | $357 | (S)-4-Benzyl-5-oxomorpholine-3-carboxylicAcid |
Buy |
| Activate Scientific AS21712 | 5G | $233 | (S)-4-Benzyl-5-oxomorpholine-3-carboxylic acid 97+% ee |
Buy |
| Chemenu CM119831 | 10g | $290 | (S)-4-Benzyl-5-oxomorpholine-3-carboxylicAcid 97% |
Buy |
(3S)-4-Benzyl-5-oxomorpholine-3-carboxylic acid Chemical Properties,Usage,Production
Synthesis
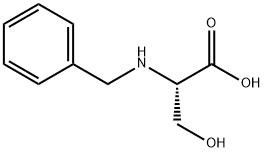
106910-76-3
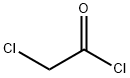
79-04-9
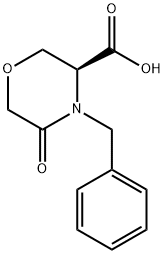
106973-37-9
General procedure: N-benzyl-L-serine (1.0 eq.) and tetrahydrofuran (THF, 6.1 vol.) were added to the reactor. The resulting solution was cooled to 0-5 °C and a pre-cooled aqueous solution of potassium carbonate (3.0 eq.) (6.1 vol., 0-5 °C) was added. Subsequently, chloroacetyl chloride (1.4 eq.) was slowly added through the addition funnel while strictly controlling the internal temperature below 5 °C. The biphasic reaction mixture was aged at 0-5 °C with stirring for about 30 min. After aging, samples were taken for HPLC analysis. If N-benzyl-L-serine residue >6 area percent is detected, chloroacetyl chloride is added additionally. Once the reaction completeness specification was satisfied, 50 wt% sodium hydroxide solution was slowly added, maintaining the internal temperature between 5-10°C until pH stabilization >13.5. The reaction was considered complete when HPLC analysis showed total intermediates <1 area percentage. The reaction mixture was warmed up to 25°C, heptane (2.03 v/v) was added, stirred rapidly for 10 minutes and left to stratify. The upper organic phase was discarded and the aqueous phase was treated again with heptane (3.04 v/v), stirred rapidly for 10 min and left to stratify, the upper organic phase was discarded. The aqueous phase was cooled to -5-0 °C and 37 wt% hydrochloric acid was slowly added, controlling the batch temperature <10 °C until pH <2. The resulting slurry was kept at -10-0 °C for at least 4 h. The slurry was then filtered through a Whatman 1 filter. The slurry was filtered through Whatman 1 filter paper or equivalent and the filter cake was washed with pre-cooled (3-7°C) water (2 x 4.57 v/v). The wet filter cake was dried under vacuum at 40-45 °C to give 1.475 kg (84.9% yield, uncorrected) of (S)-4-benzyl-5-oxo-3-morpholinecarboxylic acid.HPLC analytical conditions: retention time 1.82 min (YMC S5 ODS column, 4.6 × 50 mm, gradient elution from 10-90% aqueous methanol containing 0.2% phosphoric acid, flow rate 4 mL /min, detection wavelength 220 nm); chiral HPLC analysis conditions: retention time 7.94 min, ee value 100% (Chiralcel OJ-R column, 150 × 4.6 mm, 5 μm, eluent: methanol: 0.2% phosphoric acid aqueous solution [50:50], flow rate 1 mL/min, detection wavelength 210 nm).
References
[1] Patent: WO2005/58245, 2005, A2. Location in patent: Page/Page column 29-30[2] Patent: WO2011/111875, 2011, A1. Location in patent: Page/Page column 91
[3] Helvetica Chimica Acta, 2004, vol. 87, # 1, p. 90 - 105
[4] Journal of the Chemical Society, Perkin Transactions 1: Organic and Bio-Organic Chemistry (1972-1999), 1985, p. 2577 - 2580
Preparation Products And Raw materials
1of2
(3S)-4-Benzyl-5-oxomorpholine-3-carboxylic acid Supplier
| Supplier | Tel | Country | ProdList | Advantage | |
|---|---|---|---|---|---|
| +1-858-6993322 | info@accelachem.com | United States | 21193 | 58 | |
| 8485655694 | sales@alchempharmtech.com | United States | 63687 | 58 | |
| +86-0371-86658258 +8613203830695 |
factory@coreychem.com | China | 29794 | 58 | |
| +86-021-021-50872116 +8613122107989 |
contact@fuxinpharm.com | China | 10000 | 58 | |
| 02768782018 18771942761 |
sales01@whmonad.com | CHINA | 979 | 58 | |
| +86-0086-577-64498589 +86-15355981851 |
sales@win-winchemical.com | China | 14306 | 58 | |
| +86-0571-88938639 +8617705817739 |
info@dycnchem.com | China | 52705 | 58 | |
| +86-027-87366298 +8615927270571 |
tianyi@skbiology.cn | China | 282 | 58 | |
| +86-571-86217390; +8618058761490 |
info@gihichemicals.com | China | 49968 | 58 | |
| +undefined18986093962 | admin@konberdbio.com | China | 1378 | 58 |
View Lastest Price from (3S)-4-Benzyl-5-oxomorpholine-3-carboxylic acid manufacturers
106973-37-9, (3S)-4-Benzyl-5-oxomorpholine-3-carboxylic acidRelated Search
The What'sApp is temporarily not supported in mainland China

