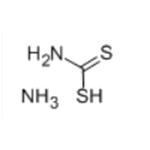
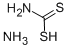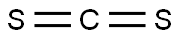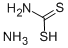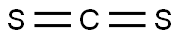Chemical Properties
Yellow crystals. Soluble in water.
Uses
Instead of H2S or (NH4)2S for pptg metals in chemical analysis; synthesis of heterocyclic compounds.
Uses
Ammonium Dithiocarbamate is an intermediate in synthesizing [6R-[6α,7β(Z)]]-7-[[(2-Amino-4-thiazolyl)(methoxyimino)acetyl]amino]-3-[[[4-(2-carboxyethyl)-2-thiazolyl]thio]methyl]-8-oxo-5-thia-1-azabicyclo[4.2.0]oct-2-ene-2-carboxylic Acid (A630540), an isomer of the antibiotic Cefodizime (C242865) which is a third generation cephalosporin with a broad spectrum of antibacterial activity.
Preparation
Ammonium dithiocarbamate is prepared most easily from ammonia and carbon disulfide in alcohol or an ester solvent.
Synthesis
GENERAL STEPS: Ammonia (1 equiv.) was passed into a solution of carbon disulfide (16.7 g, 0.22 mol) in tetrahydrofuran (THF, 100 mL) according to the literature methodology, and the reaction temperature was controlled to be below 45 °C. After 1 h of reaction, the mixture was cooled to room temperature to terminate the reaction. Excess ammonia was removed by passing nitrogen for 30 min or rotary evaporation. The precipitate was collected by filtration to afford ammonium dithiocarbamate (23.1 g, 96% yield) as a white solid. Methyl iodide (17.8 g, 0.125 mol) was added to a solution of ammonium dithiocarbamate (5.5 g, 0.05 mol) in acetone (100 mL) at room temperature and the reaction was stirred for 24 hours. The solid was collected by filtration, washed with cold acetone (20 mL) and dried under vacuum to give a white solid of S,S'-dimethyliminodithiocarbamate ammonium iodide (18.9 g, 96% yield). The S,S'-dimethyliminodithiocarbonate ammonium iodide (7.9 g, 0.02 mol) was treated with saturated sodium bicarbonate (NaHCO3) aqueous solution (100 mL × 2), and the organic product was extracted with dichloromethane (50 mL). The organic layer was dried with anhydrous sodium sulfate (Na2SO4) and concentrated in vacuum to give a clear colorless liquid product (2.3 g, 95% yield). The product was confirmed by 1H NMR (400 MHz, CDCl3) δ 8.88 (s, 1H), 2.42 (s, 6H); 13C NMR (101 MHz, CDCl3) δ 172.10, 14.50; HRMS (ESI+) m/z calculated value of C3H8N+ 122.0098, measured value 122.0096.
References
[1] Tetrahedron Letters, 2015, vol. 56, # 44, p. 6097 - 6099
[2] Journal of Medicinal Chemistry, 2010, vol. 53, # 1, p. 77 - 105
[3] Patent: TWI609015, 2017, B. Location in patent: Paragraph 0187; 0188
[4] Zeitschrift fur Anorganische und Allgemeine Chemie, 2010, vol. 636, # 2, p. 356 - 362
[5] Journal fuer Praktische Chemie (Leipzig), 1868, vol. <1>103, p. 178