Chemical Properties
white to light yellow crystal powder
Synthesis
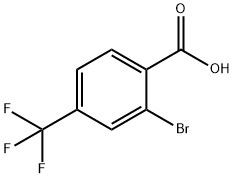
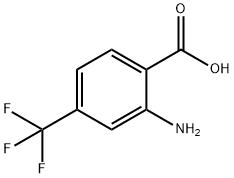
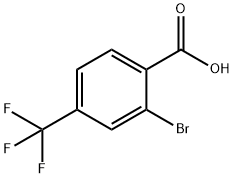
Example 5 Synthesis of 2-bromo-4-(trifluoromethyl)benzoic acid (Formula V, where R1 = H, R2 = CF3): 205 g (1 mol) of 2-amino-4-trifluoromethylbenzoic acid, 600 ml of glacial acetic acid, and 400 ml of 47% hydrobromic acid were added sequentially to a 3 liter round bottom flask. After the ingredients were completely dissolved, the reaction mixture was cooled to -10°C and diluted with 400 ml of water. Subsequently, a solution of 69 g (1 mol) of sodium nitrite dissolved in 200 ml of water was added dropwise while keeping the temperature below 0°C. After the dropwise addition, the reaction mixture was continued to be stirred at 0°C for 2 hours. The above solution was slowly added dropwise to a 6-liter reactor pre-filled with 143.5 g (1 mol) of cuprous bromide dissolved in 500 ml of 47% hydrobromic acid and maintained at 60°C. After completion of dropwise addition, the reaction mixture was stirred at 60 °C for 1 h, then cooled to room temperature and poured into two liters of ice water. The precipitated solid was filtered through a sintered glass funnel, washed with water, and dried under a stream of air to give 216 g (yield: 80%) of 2-bromo-4-(trifluoromethyl)benzoic acid as a solid with a melting point of 118.5 °C. Note: See the literature for the preparation of 2-amino-4-(trifluoromethyl)benzoic acid (formula VI, where R1 = H and R2 = CF3).
References
[1] Patent: US5514719, 1996, A
[2] Synthesis (Germany), 2012, vol. 44, # 14, p. 2173 - 2180
[3] Patent: WO2005/54176, 2005, A1. Location in patent: Page/Page column 62