Synthesis
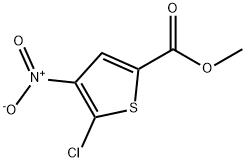
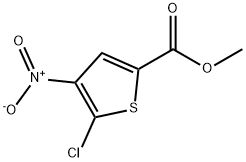
Step 2: Preparation of methyl 5-chloro-4-nitrothiophene-2-carboxylate
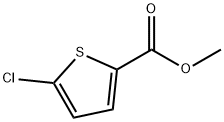
Fuming nitric acid (15 mL) was placed in an ice bath and cooled with saturated brine. Methyl 5-chlorothiophene-2-carboxylate (4.99 g, 28.3 mmol) was added slowly in small batches to avoid violent reaction. After addition, the reaction mixture was stirred in an ice bath for 1 hour. Upon completion of the reaction, the mixture was poured into ice water and a solid was precipitated. The precipitate was collected by filtration, washed with water and dried under vacuum to afford methyl 5-chloro-4-nitrothiophene-2-carboxylate (3.88 g, 58% yield). The product was characterized by 1H-NMR (400 MHz, DMSO-d6): δ 8.17 (1H, s).
References
[1] Patent: EP1688420, 2006, A1. Location in patent: Page/Page column 122
[2] Patent: CN107903208, 2018, A. Location in patent: Paragraph 0136; 0137; 0139
[3] Journal of the American Chemical Society, 1952, vol. 74, p. 2965,2969